Statements of Second Law of Thermodynamics:
Kelvin’s Statement:
It states that, “It is impossible to create a heat engine that can convert all of the heat it absorbs from a hot reservoir into useful work without losing heat to a cold reservoir”
It is applicable to heat engines. In heat engine, the working substance takes some heat from the source (at higher temperature) and converts a part of it into mechanical workdone and it rejects the remaining part to the sink (at lower temperature). So, presence of sink is essential for continuous conversion of heat into work. So, we cannot obtain continuous supply of work from single body.
Clausius Statement:
It states that, “It is impossible for a self-acting machine unaided by any external agency to convey heat from one body to another at a higher temperature. i.e., we cannot make heat flow from a colder body to a warmer body without doing external work. It is based on the performance of refrigerator, which is a heat engine working on the backward direction. In the refrigerator, the working substance takes in heat from a cold body, a certain amount of work is done on it by some outside agency and rejects a larger amount of heat to a hotter body. So, refrigerator can transfer heat from a cold body to hot body with the aid of an external agency.
Heat Engine:
It is a device used to convert heat energy into mechanical energy. The block diagram of heat engine is shown in the given figure. Heat engine consists of three parts:
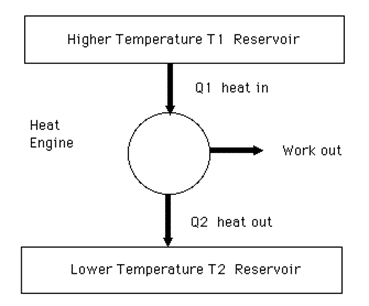
- Source
- Sink and
- Working substance
(i) Source:
Source is the reservoir of heat at high temperature T1.
(ii) Sink:
Sink is the reservoir of heat at temperature T2, when T2 < T1.
(iii) Working substance:
The substance which absorbs heat from the source & perform mechanical work & remaining heat is transferred to sink.
Efficiency of heat engine ([latex]\eta[/latex]):
It is defined as the ratio of external workdone to the heat energy absorbed by the working substance from the source. It is denoted by [latex]\eta[/latex].
i.e. [latex]\eta = \frac{External\ workdone}{heat\ energy\ absorbed\ from\ the\ source}[/latex]
Let, Q1 be the amount of heat absorbed by the working substance from the source and Q2 be that rejected to the sink. Then, the external workdone = (Q1 – Q2)
So, the efficiency of the heat engine becomes, [latex]\eta = \frac{Q_1 – Q_2}{Q_1}[/latex]
= [latex]1 – \frac{Q_2}{Q_1}[/latex]
No engine will convert all heat absorbed from the source to work. So, efficiency of heat engine is always less than 100%.
Carnot Heat Engine:
In 1824, Carnot devised an ideal cycle for heat engine. The parts of it are shown in figure below.
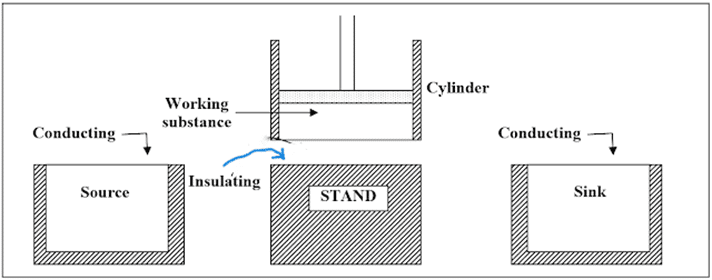
It consists of:
- Source: A hot body of infinite thermal capacity is a source, which is maintained at a fixed higher temperature T1, from which the working substance draws heat Q1.
- Cylinder: A perfectly non-conducting and having frictionless piston enclosing the ideal gas is called cylinder. The walls of it are perfect insulator while the bottom (base) is a perfect heat conductor.
- Sink: It is at a fixed lower temperature T2, to which any amount of heat can be rejected.
- Working Substance: An ideal gas acts as the working substance, inside the cylinder.
Apart from these, there is also a perfectly insulating stand where the cylinder can be placed. So, it would be completely isolated working substance. It is subjected to a cycle of four operations having two of them isothermal and two adiabatic. Such cycle is called a Carnot cycle.
Carnot Cycle:
Let us consider, n mole of an ideal gas is enclosed in the cylinder. P1, V1, T1 be its initial pressure, volume and temperature. Point A be the initial state of the gas. There are four operations in Carnot cycle. They are:
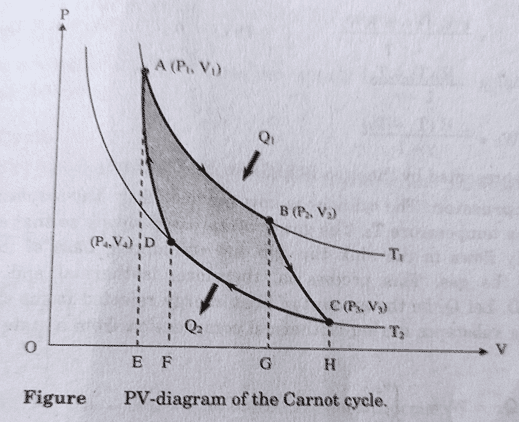
(i) Isothermal Expansion:
First, the cylinder is placed on top of source having infinite thermal heat capacity. It draws some heat from the source and the gas expands at constant temperature (T1). Thus, the process is isothermal expansion (from A to B).
So, the workdone in the process (W1 = Q1) = [latex]nRT_1 ln(\frac{V_2}{V_1})[/latex]
(ii) Adiabatic Expansion:
Now, the cylinder is placed on the insulating stand and the gas is allowed to expand further from B to C such that no heat is lost or gained to the surroundings. i.e. the process is adiabatic expansion.
Workdone in this process (W2) = [latex]\frac{nR(T_1 – T_2)}{\gamma – 1}[/latex]
(iii) Isothermal Compression:
Now, the cylinder is removed from insulating stand and placed on the heat sink at temperature T2. The gas is slowly compressed such that temperature remains T2. The gas is compressed from C to D.
So, workdone in the process (Q2 = W3) = [latex]-nRT_2 ln(\frac{V_4}{V_3}) = nRT_2 ln(\frac{V_3}{V_4})[/latex]
(iv) Adiabatic Compression:
The cylinder is again placed on the insulating stand and piston is moved downward such that adiabatic compression occurs along DA and it returns back to initial condition i.e. P1 and V1.
So, workdone during this process (W4) = – [latex]\frac{nR(T_2 – T_1)}{\gamma – 1} = \frac{nR(T_1 – T_2)}{\gamma – 1}[/latex]
Now, total workdone by the gas = W1 + W2 and,
Total workdone on the gas = W3 + W4
Now, net workdone by the engine per cycle (W) = W1 + W2 – (W3 + W4)
= W1 – W3
(Represented by the area bounded by ABCDA).
Efficiency of Carnot Cycle:
As we know,
[latex]\eta = 1 – \frac{Q_2}{Q_1}[/latex]
= [latex]1 – \frac{nRT_2 ln(\frac{V_3}{V_4})}{nRT_1 ln(\frac{V_2}{V_1})}[/latex]
= [latex]1 – \frac{T_2 ln(\frac{V_3}{V_4})}{T_1 ln(\frac{V_2}{V_1})}[/latex] ………………………………………….. (i)
Since, for A to B (isothermal): P1V1 = P2V2 ………….. (ii)
For B to C (adiabatic): [latex]P_2V_2^\gamma = P_3V_3^\gamma[/latex] ……………… (iii)
For C to D (isothermal): P3V3 =P4V4 ……………………… (iv)
For D to A (adiabatic): [latex]P_1V_1^\gamma = P_4V_4^\gamma[/latex] …………… (v)
Diving eq (iii) by (v),
[latex]\frac{P_2V_2^\gamma}{P_1V_1^\gamma} = \frac{P_3V_3^\gamma}{P_4V_4^\gamma}[/latex]
Or, [latex]\frac{V_1V_2^\gamma}{V_2V_1^\gamma} = \frac{V_4V_3^\gamma}{V_3V_4^\gamma}[/latex]
Or, [latex](\frac{V_2}{V_1})^{\gamma – 1} = (\frac{V_3}{V_4})^{\gamma – 1}[/latex]
Or, [latex]\frac{V_2}{V_1} = \frac{V_3}{V_4}[/latex]
So, eq (i) becomes,
Or, [latex]\eta = 1 – \frac{T_2 ln(\frac{V_3}{V_4})}{T_1 ln(\frac{V_3}{V_4})}[/latex]
Or, [latex]\eta = 1 – \frac{T_2}{T_1}[/latex]
In Percentage, [latex]\eta = (1 – \frac{T_2}{T_1})\times 100%[/latex]
This is the required expression for efficiency of carnot cycle.
Petrol Engine:
It was developed by Otto in 1876 so it is also called Otto engine. It consists of a cylinder fitted with the piston. There is inlet and outlet valve named I and O as shown in figure inside the cylinder. It is four stroke engine which uses a mixture of petrol and air (98% of air and 2% of petrol). The working of this engine is described as follows:
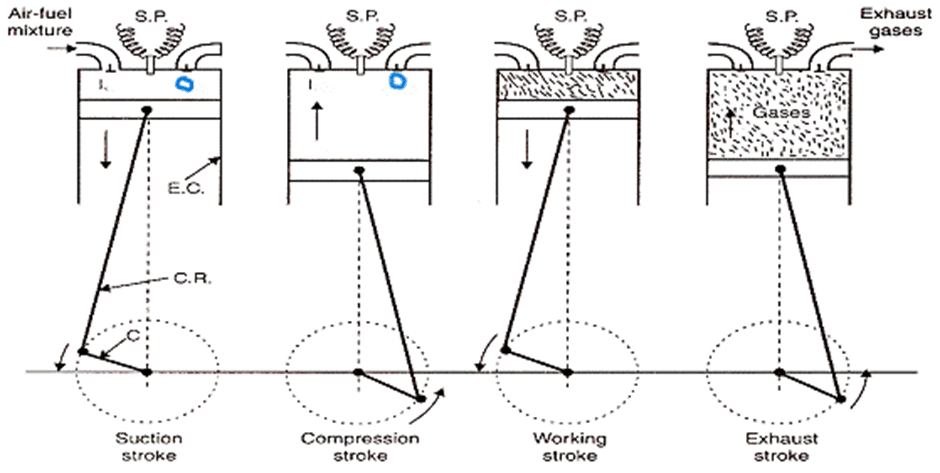
- Suction stroke: In this stroke, the inlet valve I is opened and the mixture of air and petrol is sucked in by the outward motion of the piston. It is represented by the line AB in PV diagram.
- Compression stroke: In this stroke, both inlet I and outlet O valve is closed and the mixture undergoes adiabatic compression such that its volume is decreased about 1/5th of the original volume, which increases the temperature upto 600oC. It is represented by the curve BC in PV diagram.
- Working Stroke: In this stoke, inlet and outlet valve are closed. A spark is produced and the mixture of petrol and air ignites, which rises the temperature about 2000oC and pressure of 15 atm is developed. Here, Q1 amount of heat is generated at constant volume represented by the line CD. The gas expands adiabatically and working stroke is represented by DE.
- Exhaust Stroke: The outlet valve opens and burnt gas are exhausted out to the atmosphere. It is represented by the line BA in PV diagram. The four stroke processes are shown in figure above.
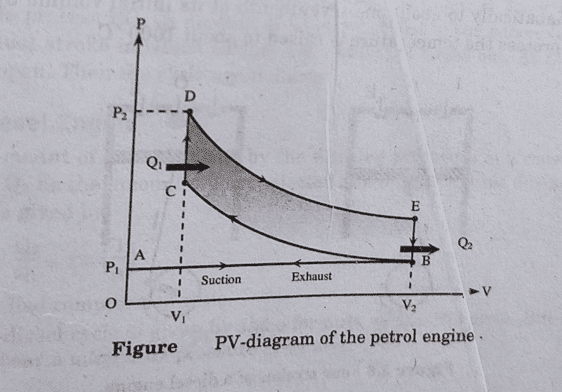
After the four strokes, the engine is ready for new fresh cycle. The PV diagram is shown alongside.
Efficiency of petrol engine:
If Q1 be the amount of heat taken and Q2 be the amount of heat rejected from the engine, then, workdone by the engine is given by
W = Q1 – Q2
The efficiency of petrol engine is ([latex]\eta[/latex]) = [latex](1 – \frac{Q_2}{Q_1})\times 100%[/latex]
For adiabatic expansion, [latex]T_1V_1^{\gamma – 1} = T_2V_2^{\gamma – 1}[/latex]
Or, [latex]\frac{T_2}{T_1} = (\frac{V_1}{V_2})^{\gamma – 1}[/latex]
But, efficiency,
[latex]\eta = 1 – \frac{T_2}{T_1}[/latex]
= [latex]1 – (\frac{V_1}{V_2})^{\gamma – 1}[/latex]
[latex]\eta = 1 – (\frac{1}{\rho})^{\gamma – 1}[/latex]
Where, [latex]\rho = \frac{V_2}{V_1}[/latex] is called compression ratio and [latex]\gamma[/latex] is the ratio of two specific heats of gas.
Merits and Demerits of petrol engine:
Merits:
- It is useful to cars, bikes, scooters, etc.
- It is small in size and also light weight.
- Its efficiency is more than the steam engine.
- It is practically smokeless.
Demerits:
- The cost of petrol is high, which causes this more expensive.
- Due to its explosive nature, there is always a risk of fire.
Diesel Engine
Diesel engine was developed by Rudolf Diesel which consists of cylinder fitted with movable piston. There are three holes in a cylinder fitted with inlet valve (I), outlet valve (E) and oil spray (O). The piston of diesel engine connected to a wheel through the rod. The working of diesel engine completes in 4 steps.
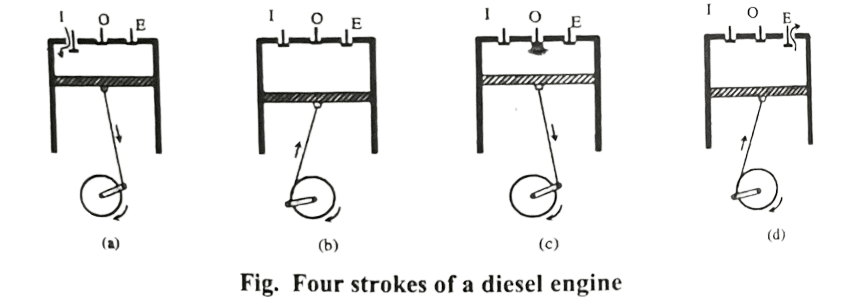
- Suction Stroke: In this stroke, air valve I is opened and pure air is sucked in at atmospheric pressure. EC represents the suction stroke in PV diagram.
- Compression Stroke: In this stroke, the piston starts moving inwards and air is compressed adiabatically to 1/17th of its initial volume, which rises the temperature to about 1000oC. CD represents the adiabatic compression stroke in PV diagram.
- Working Stroke: In this stroke, the oil valve O is opened and diesel oil is forced in. At high temperature, the oil burns. The supply of oil is so regulated that the pressure remains constant. Here, burning fuel supplied some heat Q1 to the air at constant pressure. At a certain stage, the temperature reached to 2000oC, the oil supply is cut off. In PV diagram, DA represents isobaric burning of oil. At A, supply of oil is cut off. AB represents the adiabatic expansion and volume becomes V1.
- Exhaust Stroke: In this stroke, exhaust valve E is opened and the useless gas mixture is forced out of the cylinder and the engine is ready for new fresh cycle. At B, exhaust valve is opened and pressure drops to C. Here, Q2 amount of heat is rejected to the surroundings. CE represents the exhaust stroke, where useless gas mixture is forced out.
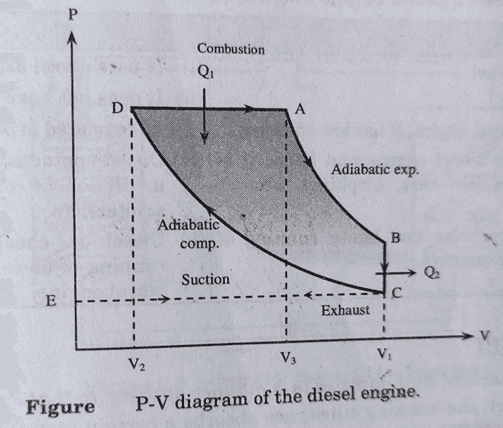
Efficiency of Diesel Engine:
Let, Q1 be the amount of heat absorbed by the working substance and Q2 be that rejected to the surroundings, then, the efficiency of diesel engine is given by:
[latex]\eta = 1 – \frac{Q_2}{Q_1} = 1 – (\frac{1}{\rho})^{\gamma – 1}[/latex]
Where, [latex]\rho = \frac{V_1}{V_2}[/latex] is called compression ratio.
The efficiency of diesel engine is given by the same formula of otto cycle.
Merits and demerits of diesel engine:
Merits:
- It has higher efficiency than that of petrol engine.
- It is less inflammable than petrol engine.
- Diesel oil is cheaper than petrol oil.
Demerits:
It is heavier than petrol engine. Due to this, we cannot use this engine in lighter vehicles like bike, scooters, etc.
Refrigerator:
A device that operates in a manner opposite to that of a heat engine is called refrigerator. In refrigerator, the working substance takes some heat Q2 from the sink at T2. The quantity of heat delivered to the source is Q1 = Q2 + W, where, W is the external workdone on the working substance. Thus, the refrigerator transfers heat from cold body to hot body when some external workdone is performed on the system.
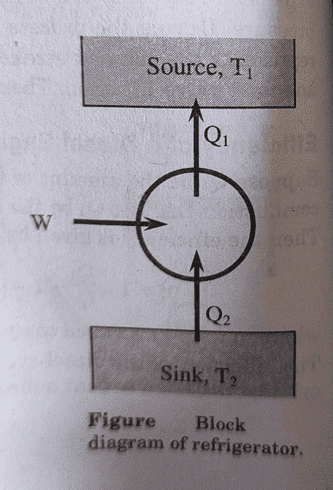
Coefficient of Performance of Refrigerator:
It is defined as the ratio of amount of heat taken from the cold body (sink) to the work done in running the machine. It is denoted by [latex]\beta[/latex].
i.e.
[latex]\beta = \frac{Q_2}{W} = \frac{Q_2}{Q_1 – Q_2}[/latex]
= [latex]\frac{\frac{Q_2}{Q_1}}{1 – \frac{Q_2}{Q_1}}[/latex]
= [latex]\frac{\frac{T_2}{T_1}}{1 – \frac{T_2}{T_1}}[/latex] [latex][∵ \frac{Q_2}{Q_1} = \frac{T_2}{T_1}][/latex]
= [latex]\frac{T_2}{T_1 – T_2}[/latex]
The smaller the value of T1 – T2, the larger will be the coefficient of performance of the refrigerator.
Working:
The figure above shows the schematic diagram of refrigerator. The working substance used is called refrigerant eg. Frozen, ammonia, SO2, etc. In the diagram, P is the compressor in which the working substance is compressed to a high pressure. The hot high-pressure gas coming out through the valve V2 is cooled by passing through the spiral pipe S in the condenser, where cold water is circulated.
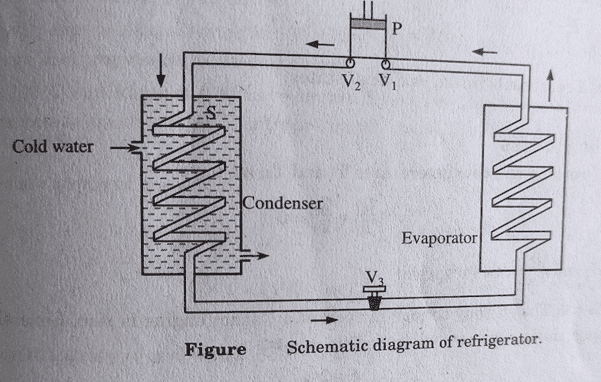
The compressed vapor gets liquified. Through a throttling valve V3, the high-pressure liquid enters the spiral tube in the evaporator. The necessary heat is taken from the materials kept inside the evaporator. The vapor coming out of the evaporator is sucked back into the compressor through the valve V1. And, the process is repeated. So, continuous cooling is produced.
Entropy and Disorder:
Entropy is such a property of the state of a system as temperature, pressure, volume and internal energy. It measures the degree of disorder or randomness in a system.
If dQ is the amount of heat added to the gas at constant temperature, then, from first law of thermodynamics,
dQ = dW [U = constant for isothermal process, dU = 0]
= PdV = [latex]\frac{nRT}{V}dV[/latex]
Or, [latex]\frac{dV}{V}=\frac{dQ}{nRT}[/latex]
We define entropy change of a system dS at temperature T as,
dS = [latex]\frac{dQ}{T}[/latex]
It is just measure of increase of disorder.