No. of electron = no. of proton in all atoms
Charge on electron = [latex]1.6\times 10^{-19}[/latex] C and is denoted by e.
Mass of electron is denoted by me and is equal to [latex]9.1\times 10^{-31}[/latex] Kg.
Millikan’s Oil Drop Experiment
To measure the charge of an electron, an experiment was performed on 1909 by Millikan, known as the Millikan’s oil drop experiment. The experimental arrangement is shown in figure alongside. It consists of two metal plates A and B with diameter 20 cm. It has a small hole at the upper plate A. The upper plate is connected to a high-tension battery and the lower plate is earthed. It is then enclosed by double walled chamber.
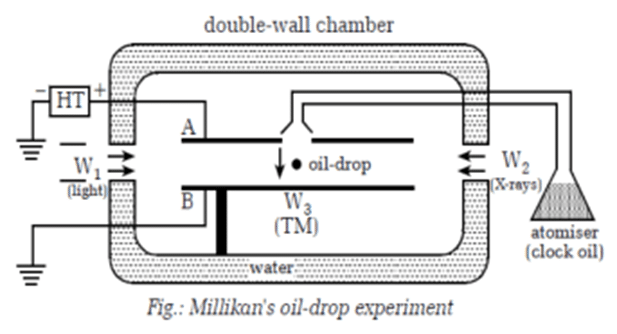
The clock oil is sprayed through the upper plate through hole with the help of atomizer. Due to friction, the drops get charged and acquire a few electronic charges. Light is passed through window W1 to illuminate the oil drops. If the oil drops are not ionized by friction, x-rays are passed through window W2. The motion of the drops is observed by microscope.
Theory 1: Motion of oil drop under gravity alone
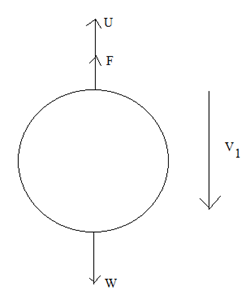
Let us suppose, at first, no electric field is applied, when the oil drop moves under gravity alone, it first increases its velocity. After some time, the oil drops move with constant velocity, called terminal velocity.
Let, r = radius of oil drop.
m = mass of oil drop
[latex]\rho[/latex] = density of oil
[latex]\sigma[/latex] = density of air
Then,
Vol. of the oil drop = [latex]\frac{4}{3}\pi r^3[/latex]
Its weight (w) = mg
= [latex]\rho[/latex]vg
= [latex]\frac{4}{3}\pi r^3\rho g[/latex]
\Upthrust due to air (U) = wt. of air displaced by drop
= [latex]\frac{4}{3}\pi r^3 \sigma g[/latex]
\The viscous force acting upward on the drop (F) = 6[latex]\pi \eta rv_1[/latex]
When the oil drop is moving with terminal velocity [latex]v_1[/latex], then,
F+U = W
[latex]\therefore[/latex] F = W – U
[latex]6\pi \eta rv_1 = \frac{4}{3}\pi r^3\rho g – \frac{4}{3}\pi r^3 \rho g[/latex]
[latex]\therefore r = \sqrt{\frac{9\eta v_1}{2(\rho – \sigma)g}}[/latex] …………….. (i)
By knowing the values of [latex]\eta, v_1, \rho, \sigma[/latex] and g, radius of the oil drop can be calculated.
Theory 2: Motion of the oil drop under electric field
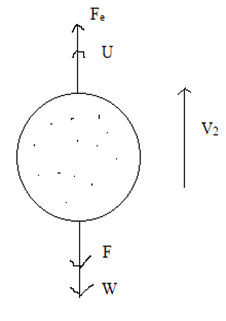
Let, a strong electric field is applied between the plates such that the force due to electric field acts in the vertically upward direction.
Now, the oil drop moves in upward direction and attains a terminal velocity [latex]v_2[/latex] in upward direction. Let, E be the electric field intensity, then, electrostatic force on the drop of charge q is given by:
Fe = qE
Viscous force (F) = 6[latex]\pi \eta rv_2[/latex]
Now,
Fe + U = F+ W
[latex]\therefore[/latex] Fe = (W – U) + F
qE = [latex]\frac{4}{3}\pi r^3(\rho – \sigma)g + 6\pi \eta rv_2[/latex]
Or, qE = [latex]6\pi \eta rv_1 + 6\pi \eta rv_2[/latex]
[latex]\therefore q = 6\pi \eta \frac{(v_1 + v_2)}{E}\times \sqrt{\frac{9\eta v_1}{2(\rho – \sigma)g}}[/latex] …………………. (ii)
This is the required expression for charge on the oil drop.
If the oil drop moves with constant terminal velocity downward even if the electric field is applied, then, the viscous force will act upward and the charge on the oil drop will be:
[latex]\therefore q = 6\pi \eta \frac{(v_1 – v_2)}{E}\times \sqrt{\frac{9\eta v_1}{2(\rho – \sigma)g}}[/latex]
The experiment was repeated several times and the result was consistent and found to be equal to the integral multiple of [latex]1.6\times 10^{-19}C[/latex].
Cathode rays
When the pressure inside the discharge tube is kept 0.01 mm of Hg, an invisible ray emerges out of the cathode, called cathode rays.
Properties
- It is emitted normally from the surface of cathode.
- It can penetrate small thickness of matter like aluminium foil.
- It can pass in straight line.
- As they carry negative charges, they can be deflected by electric and magnetic fields.
- They have some momentum and K.E.
- When light falls on matter, they produce heat.
- The velocity of cathode rays are [latex]\frac{1}{10}^{th}[/latex] of velocity of light.
- It can produce x-rays if cathode rays are suddenly stopped.
Motion of electron beam in electric fields
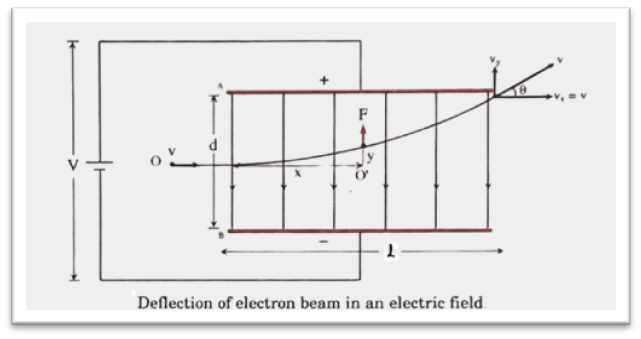
Let, a p.d. of V be applied between two parallel plates A and B separated by a distance of d so that the upper plate A is positively charged and the lower plate B is negatively charged as shown in the figure. E = [latex]\frac{V}{d}[/latex] is the magnitude of electric field intensity in between the plates. [latex]l[/latex] is the length of either plate A or B. For simplicity, a horizontal line OO‘ that passes through the middle of the distance between A and B, is taken as x-axis.
Let, v be the velocity of an electron having charge (e) and mass (m) just before entering the electric field (i.e., at the point O). It moves along the direction shown in figure in the field and then goes out of the field alone the straight-line.
Let, P(x, y) be the position of the electron at any instant, considering its horizontal motion, we have,
x = horizontal distance = vt………………………………….. (1)
Considering its vertical motion, we have,
Y = ut+ [latex]\frac{1}{2}at^2[/latex]
= 0 + [latex]\frac{1}{2}at^2[/latex] [[latex]\because[/latex] at the point O, for vertical motion, u = 0]
= [latex]\frac{1}{2}\frac{F}{m}t^2[/latex] [∵ F = ma]
= [latex]\frac{1}{2}\frac{eE}{m}t^2[/latex] [∵ F = eE]
= [latex]\frac{1}{2}\frac{eE}{m}(\frac{x}{v})^2[/latex] [using eqn (1)]
Or, y = [latex](\frac{eE}{2mv^2})x^2[/latex]
[latex]\therefore y = (\frac{1}{2}\frac{eV}{mdv^2})x^2[/latex] ……………… (2)
Here, e and m are constant for an electron. If E and v are constant, the equation (2) is in the form of eqn of parabola. (i.e., y = ax2). Hence, the path of an electron is parabola in electric field.
The component of the velocity [latex]v_y[/latex] is given by,
[latex]v_y = [/latex] acceleration [latex]\times[/latex] time = [latex]\frac{eV}{md}\times \frac{l}{v}[/latex] [[latex]\because[/latex] when electron just passes the plates, [latex]x = l[/latex]]
So, the magnitude of the velocity (v) of the beam will be,
[latex]v = \sqrt{v_x^2 + v_y^2}[/latex]
Also, the angle at which the beam emerges out from the field is given by,
[latex]tan\theta = \frac{v_y}{v_x} = \frac{eV}{md}\times \frac{l}{v} = \frac{eVD}{dmv^2}[/latex]
Motion of electron beam in magnetic field
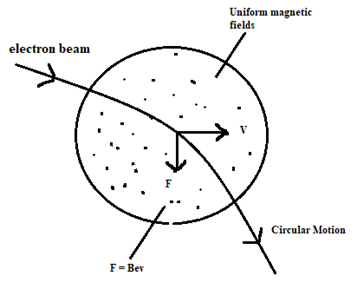
Let, an electric beam, moving with a speed v, enters a uniform magnetic field of magnitude B acting perpendicular to the direction of motion, as shown in figure. The force (F) on the electron is then given by:
F = Bev…………………………. (1)
The direction of force is perpendicular to both B and v. The force is always normal to the path of the electron beam. If the field is uniform, the force is constant in magnitude because it deflects the electron but does not change its speed (v). The beam travels in a circular path of radius (r). In this case, F = Centripetal force.
[latex]\therefore Bev = \frac{mv^2}{r}[/latex], where, m is the mass of the electron.
[latex]\therefore r = \frac{mv}{Be}[/latex], which gives the radius of the circular path.
Now, Be = [latex]\frac{mv}{r}[/latex]
Or, Be = m[latex]\omega[/latex] [∵v = [latex]\omega[/latex]r]
Or, Be = m.[latex]\frac{2\pi}{T}[/latex]
[latex]\therefore T = \frac{2\pi m}{Be}[/latex] ……………………………………. (2)
Equation (2) shows that the time period (T) is independent of speed of the electron (v).
Specific charge of an electron
The ratio of charge and mass of an electron is called the specific charge of an electron. Since, the mass of the electron is m = 9.1×10-31 Kg and the charge on it is: e = 1.6×10-19 C, its specific charge is given by:
Specific charge = [latex]\frac{Charge\ on\ the\ electron\ (e)}{mass\ of\ the\ electron\ (m)}[/latex]
= [latex]\frac{1.6\times 10^{-19}}{9.1\times 10^{-31}}[/latex]
[latex]= 1.76\times 10^{11} C/Kg[/latex].
J.J. Thomson’s method to determine the specific charge on an electron
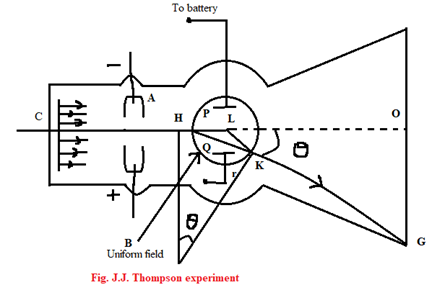
In the apparatus, C and A are cathode and anode respectively. Cathode rays pass through the narrow-slit cut at A. The rays then strike the glass at O. The rays can be deflected electrostatically by applying potential difference across the two plates PQ of magnetically by means of a current passing through two Helmholtz coils on either side of the tube near P and Q. The magnetic field B is perpendicular to the paper and if it is uniform, a constant force acts on the cathode rats (electrons) normal to its motion of electrons. They thus begin to move alone the arc HM of a circle of radius r and they move in a straight-line MG when they leave the field.
So, force (F) = Bev = [latex]\frac{mv^2}{r}[/latex].
Where, e is the charge on an electron and M is its mass.
Or, Be = [latex]\frac{mv}{r}[/latex]
Or, [latex]\frac{e}{m} = \frac{v}{rB}[/latex] ………………… (1)
Now, from the figure, we have, [latex]\theta \approx tan\theta = \frac{OG}{OL}[/latex] ……………….. (2)
And, [latex]\theta = \frac{HK}{r}[/latex] ………………….. (3) [∵ [latex]\theta = \frac{l}{r}[/latex]]
From (2) and (3), we have,
[latex]\frac{OG}{OL} = \frac{HK}{r}[/latex]
Or, r = [latex]\frac{HK.OL}{OG}[/latex] ……………… (4)
Here, OG is the deflection of cathode beam due to the magnetic field. It can be measured. OL is the distance between the centre of the magnetic field and screen. HK is the length of magnetic field. Thus, Knowing HK, OL can be found.
Now, the electric field E between P and Q is varied until the beam was returned to the point O. Since there is no deflection in the electron beam, electric and magnetic fields are perpendicular to each other. In this condition, we have,
eE = Bev
Or, v = E/B ………………. (5) Substituting the values of r and v from equations (4) and (5) in eqn (1), the specific charge of electron is determined and that is found to be 1.76[latex]\times[/latex]1011 C/Kg.