Introduction
The radiation is emitted from a black body in separate packets of energy, each packet of energy is called quantum of energy. Each quanta carries a definite amount of energy called photon and is given by:
Photon energy, E = hf, where f is the frequency of radiation, h is a constant called planks constant and is equal to 6.62×10-34 Js.
This theory is called quantum theory of radiation.
Photoelectric effect
When radiation of suitable frequency such as x-rays, ultraviolet and infrared light is made to fall on a metal surface, it emits electron from the surface. This phenomenon is known as photoelectric effect and the emitted electrons are called photoelectrons.
Properties of photons
- Photons travels in a vacuum with the speed of light i.e., 3×108 m/s.
- Each photon has energy hf and a definite linear momentum equal to hf/c or h/[latex]\lambda[/latex].
- A photon may collide with a material particle. This collision is considered as perfectly elastic collision where total energy and total momentum are conserved.
- The photons are electrically neutral. Hence, they are not affected by electric and magnetic fields.
Experimental study of photoelectric effect
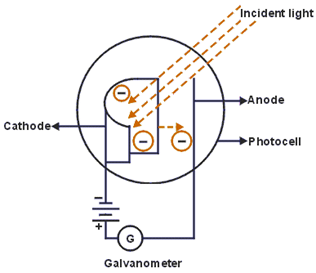
The figure alongside is the experimental setup for the study of photoelectric effect. It consists of two metal plates C for cathode and A for anode. With the help of battery, a constant potential difference is maintained between them. It was then enclosed by an evacuated tube so as to prevent it from oxidation by air.
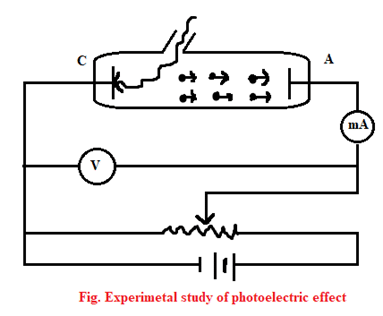
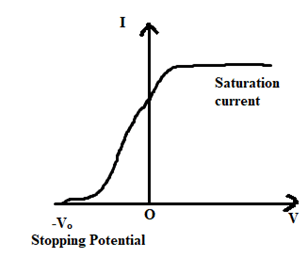
When a radiation of suitable frequency falls on C, electrons are emitted and gets accelerated towards A, if it is kept at positive potential with respect to the cathode. Then, a photoelectric current flow outside the circuit for a fixed frequency and intensity of incident light, the photoelectric current then increases with the applied positive potential until it attains a maximum value. When all the photoelectrons get emitted from the cathode (C), the maximum current is called the saturation current.
Now, the potential of plate A is made negative by gradually decreasing it, with respect to cathode (C). We now find that photoelectric current decreases rapidly and becomes zero. The minimum value of negative potential to the plate A at which the photoelectric current becomes zero is called the stopping potential (Vo). In such case, the workdone by the stopping potential is equal to the maximum K.E. of the photoelectrons emitted. i.e.
[latex]eV_o = \frac{1}{2}mv_{max}^2[/latex],
where, vmax = maximum velocity of the photoelectrons emitted.
Threshold frequency
There is a certain minimum frequency of radiation below which there is no photoelectric emission whatever be the intensity of radiation. This minimum frequency is called threshold frequency. Different metals have different threshold frequency. It is denoted by fo.
Work function
The minimum amount of energy required to eject an electron just from the surface of a metal is called the work function of the metal. It is constant for a given metal but it differs from metal to metal. It is denoted by [latex]\phi[/latex] and is given as:
[latex]\phi[/latex] = hfo, where, h = Planck’s constant.
fo = threshold frequency.
Einstein’s photoelectric equation
In 1905, Einstein explained the phenomenon of photoelectric effect on the basis of Planck’s quantum theory of radiation.
The energy associated with each particle or photon of light with frequency n is: E = hf
When a photon falls on a metal, it transfers the whole amount of energy hn to any one of the electrons present in the metal. The energy hn is utilized in two ways: –
- A part of this energy is used in ejecting the electrons from the metal. The minimum amount of energy necessary to eject an electron from metal surface is called work function of the metal.
- The rest of the energy is used in providing the K.E. to eject electrons.
Let, a photon of energy hf is incident on a metal having work function [latex]\phi[/latex] and the K.E. of ejected electron is Ek, then,
[latex]hf = \phi + E_k[/latex]
If m be the mass of the electron with velocity v, then,
[latex]hf = \phi + \frac{1}{2}mv^2[/latex] ……………………………….. (1)
This equation is called Einstein’s photoelectric equation.
If fo be the threshold frequency of light, then, work function,
[latex]\phi = hf_o[/latex]
Then, eqn (1) becomes,
[latex]hf = hf_o + \frac{1}{2}mv^2[/latex]
[latex]\therefore \frac{1}{2}mv^2 = h(f-f_o)[/latex]
Thus,
- The K.E. of emitted electrons increases with frequency of incident radiation.
- There should be a threshold frequency fo, below which no electrons are ejected.
Experimental Verification of Einstein’s photoelectric equation
A simplified from of Millikan’s apparatus is shown in figure. T is an evacuated glass tube inside which are placed three alkali metals Lithium (Li), Sodium (Na) & potassium. A wheel W can be rotated to bring the appropriate metal facing light. The metals are kept at positive potential w.r.t a gauge cylinder, which collects photo-elements reaching it. The electrometer E measures the current.
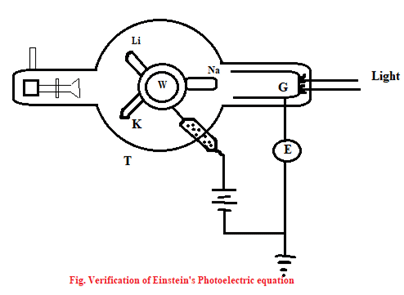
When a beam of monochromatic light falls on a target metal (say Na), the photoelectron is emitted, which then experience a retarding p.d. as the gauge cylinder G is at negative potential w.r.t. the target metal. The p.d. (V) between G and Na is increased negatively until the current becomes zero. Such p.d. is called stopping potential (Vo) for which photoelectrons with the maximum K.E. are unable to just reach the cylinder (G).
If [latex]\frac{1}{2}mv_{max}^2[/latex] is the K.E. of the fastest photoelectrons & Vo be the stopping potential, then,
[latex]\frac{1}{2}mv_{max}^2 = eV_o[/latex] ………………………. (1)
Let us consider, Einstein’s photoelectric equation,
[latex]hf = hf_o + \frac{1}{2}mv_{max}^2[/latex]
[latex]\therefore \frac{1}{2}mv_{max}^2 = hf – hf_o[/latex] ………………… (2)
From eqn. (1) & (2), we get,
eVo = [latex]hf – hf_o[/latex]
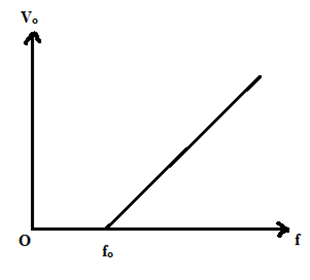
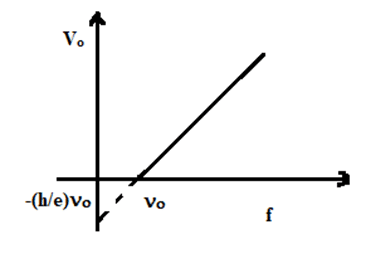
Or, Vo = [latex]\frac{h}{e}f + (-\frac{h}{e})f_o[/latex] …………………………. (3)
This eqn. (3) is in the form of y = mx + c, which is the equation of straight-line having slope h/e & y – intercept [latex](-\frac{h}{e})f_o[/latex]. A graph plotted between frequency of incident light (f) & stopping potential (Vo) is shown alongside.
This experimental graph is in agreement with Einstein’s photoelectric equation. It is clear that the slope (h/e) multiplied by electronic charge (e) gives the value of Planck’s constant (h). Millikan repeated the experiment with the other metals also & got fairly the same value of Planck’s constant i.e., 6.62 [latex]\times 10^{-34}\ Js[/latex]. This confirmed Einstein’s photoelectric theory.
Uses of photoelectric effect
Photoelectric cells are one of the major uses of photoelectric effect. There are three types of photoelectric cells.
- Photo emissive cells.
- Photo-voltaic cells and
- Photo-conductive cells
- Photo-emissive cells
It consists of an evacuated glass inside which there is two electrodes C & A. Cathode is semi-cylindrical metal having lower work function. A p.d. of 100V is applied to the anode. When light of suitable frequency falls on the cathode C, photoelectrons are emitted & a small current flows through it. It can thus record rapid changes in the intensity of light. So, it can be used in televisions.
- Photo – Voltaic cells
Into a copper plate, a thin semiconductor layer is deposited. On this, a thin film of silver or gold is deposited. When light falls on it, it penetrates the film & electrons are ejected & flows through the external low resistor (R).
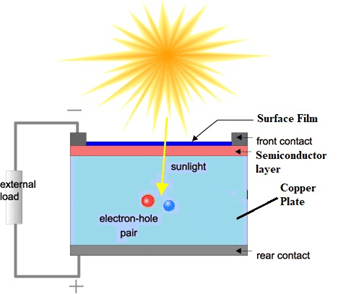
Advantages of photo-voltaic cells
- No need of external battery.
- Cheap cell &
- It is solid or strong cell.
- Photo-conductive cell
When the electrical resistance of semiconductor decreases with the increase in intensity of light, this cell works. A selenium film is deposited on the iron plate. When light of different intensity falls on the plate, a current flow through it, which contains a galvanometer & battery. Solar cell works on this principle. A picture of this cell is shown in figure alongside.
Applications of photoelectric cells
- They are used in street lights to automatically switch on or off the lights.
- It is used in cinematography for reproduction of sound.
- They are used in television camera.
- They can be used in scientific research as they are highly sensitive.
- In order to control the temperature of furnace & chemical reactions, they can be used.
Finished studying this chapter resource?
Mark this chapter as complete to update your course progress.