Internal Energy:
All materials contain molecules which are in random motion. So, they possess K.E. Also, the molecules have intermolecular force of attraction between them. So, they possess P.E. also.
The sum of kinetic energy & potential energy of a system is called its internal energy. The change in internal energy is denoted by [latex]\Delta U[/latex].
[latex]\Delta[/latex]U = K.E. + P.E.
[latex]\Delta[/latex]U = Uf – Ui
But, according to Kinetic theory of gases, the molecules of an ideal gas do not exert intermolecular forces. So, they do not possess potential energy. As a result, the internal energy of an ideal gas is wholly K.E. only and is function of temperature only.
It depends upon temperature. i.e., [latex]\Delta[/latex]U ∝ T, it is directly proportional to the temperature. It increases with increase in temperature & vice-versa.
Workdone by expansion & compression of gas:
Consider, a gas molecule is contained in a container fitted with movable piston having pressure (P), volume (V) & temperature (T) as shown in the given figure. We have,
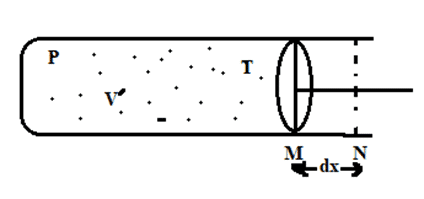
P = F/A
Force acting on piston, F = P.A
Now, let us displace piston from position M to N by very small distance dx,
Work (dW) = F. dx
= P. A. dx
dW = P. dV. [∵dV = A. dx]
Total workdone, W = [latex]\int{dw}[/latex]
= [latex]\int_{V_1}^{V_2} PdV[/latex] = P[latex]\int_{V_1}^{V_2}dV[/latex]
W = P (V2 – V1) is the workdone when gas expands from P1 to P2.
During expansion of gas, the volume V2 > V1 such that the change in volume (V2 – V1) is positive. Hence, during expansion, the work done by a system is taken as positive.
While during compression of gas, the volume V2 < V1 such that the change in volume (V2 – V1) is negative. Hence, during compression, the work done on a system is taken as negative.
First law of thermodynamics:
It is also known as principle of conservation of energy. i.e., energy can neither be created nor be destroyed, but can change from one form to another forms.
First law of thermodynamics can also be stated as, “when some quantity of heat is supplied to a system, a part of it may be used in doing external work on expansion and the rest of the heat is used to increase internal energy of the system.”
Let, dQ be the quantity of heat supplied to a system, dW be the amount of external workdone and increase in internal energy of the molecules be dU, then, from the first law of thermodynamics,
dQ = dU + dW
Since, dW = PdV, so, dQ = dU + PdV
These equations are called mathematical form of first law of thermodynamics.
If heat is given to a system, dQ = +ve
If heat is lost by the system, dQ = -ve
If work is done by the system, dW = +ve
If work is done on the system, dW = -ve
Suppose, a gas contained in a cylinder fitted with movable piston is heated at constant volume, in that case, dV = 0. So, above equation becomes,
dQ = dU + PdV = dU + 0
Or, dQ = dU. i.e., all heat supplied to a system is used to increase the internal energy.
For a cyclic process, dU = 0 because the system is brought back to initial condition. In that case,
dQ = dU + PdV = 0 + PdV
Or dQ = dW. i.e., all heat supplied to a system is used to do external work.
Real life examples:
Humans convert chemical energy in food, into K.E. by walking, running, etc.
An electric bulb converts electrical energy into light and heat energy.
Specific Heat Capacity of a Gas:
There are two types of specific heat capacity of a gas:
(a) Principal specific heat capacity at constant pressure (cp):
It is defined as the amount of heat required to raise the temperature of unit mass of a gas through unit temperature difference at constant pressure. Its SI unit is Jkg-1K-1. Mathematically,
[latex]c_p = \frac{dQ}{mdT}[/latex]
(b) Principal specific heat capacity at constant volume (cv):
It is defined as the amount of heat required to raise the temperature of unit mass of a gas through unit temperature difference at constant volume. Its SI unit is Jkg-1K-1. Mathematically,
[latex]c_v = \frac{dQ}{mdT}[/latex]
(c) Molar specific heat capacity at constant pressure (Cp):
It is defined as the amount of heat required to raise the temperature of unit mole of a gas through unit temperature difference at constant pressure. Its SI unit is Jmol-1K-1. Mathematically,
Cp = [latex]\frac{dQ}{ndT}[/latex]
(d) Molar heat capacity at constant volume (Cv):
It is defined as the amount of heat required to raise the temperature of unit mole of a gas through unit temperature difference at constant volume. Its SI unit is Jmol-1K-1. Mathematically,
Cv = [latex]\frac{dQ}{ndT}[/latex]
Relation Between Two Specific Heat Capacities of Gas (Cp – Cv = R)
Consider, n mole of ideal gas is contained in a cylinder fitted with movable piston having pressure (P), volume (V) & temperature (T) as shown in the figure.
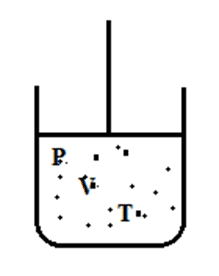
Now, let us heat this gas at constant volume through dT rise in temperature. Then, from first law of thermodynamics,
dQ = dU + PdV
nCvdT = dU + 0 [∵PdV = P.0 = 0]
nCvdT = dU ……………….. (i) where, [latex]C_v[/latex] = Molar specific heat capacity of gas
Now, let us heat the gas at constant pressure. Then, we have, from the first law of thermodynamics,
nCpdT = dU + PdV …………………… (ii) where, [latex]C_p[/latex] = Molar specific heat capacity of gas
From eqns. (1) & (2), we get,
nCpdT – nCvdT = dU + PdV – dU
∴ nCpdT – nCvdT = PdV ………………….. (iii)
For n mole of ideal gas,
PV = nRT
Differentiating both sides,
d(PV) = d(nRT)
Or, P.dV + V.dP = nR.dT
Or, PdV = nR.dT [∵VdP = dP.0 = 0]
Or, PdV = nRdT
So, eqn. (iii) becomes,
nCpdT – nCvdT = nRdT
[latex]C_p – C_v = R[/latex]
This formula is known as the Mayer’s formula.
If M is the molar mass of the gas, then eqn. (iv) can be written as,
[latex]\frac{C_p}{M} – \frac{C_v}{M} = R[/latex]
cp – cv = r, where, [latex]r = \frac{R}{M}[/latex] is the gas constant per unit mass.
Thermodynamics Process:
(i) Isothermal Process:
The process in which there is change in pressure & volume but at constant temperature, is called isothermal process.
(ii) Adiabatic Process:
The process in which there is change in pressure & volume without gain or loss of heat is called adiabatic process.
(iii) Isobaric Process:
The process in which pressure remains constant but temperature & volume changes is called isobaric process.
(iv) Isochoric Process:
The process in which volume remains constant, but pressure & temperature changes, is called isochoric process.
the gas constant per unit mass.
Workdone in isothermal process:
Consider ‘n’ molecules of gas is contained in a cylinder fitted with movable piston & have pressure, volume & temperature.
Then, workdone by gas to expand by volume dV:
dW = PdV ………………………. (i)
Total workdone by gas:
W = [latex]\int_{V_1}^{V_2}dW[/latex]
= [latex]\int_{V_1}^{V_2}PdV[/latex]
For isothermal process,
PV = nRT
P = [latex]\frac{nRT}{V}[/latex]
∴ W = [latex]\int_{V_1}^{V_2}\frac{nRT}{V}dV[/latex]
= [latex]nRT\int_{V_1}^{V_2}\frac{dV}{V} = nRT[log]_{V_1}^{V_2}[/latex]
W = nRT [logV2 – logV1]
W = nRT. log[latex]\frac{V_2}{V_1}[/latex] ………………………………….. (ii)
We have,
From Boyle’s law,
P1V1 = P2V2
∴ [latex]\frac{V_2}{V_1}=\frac{P_1}{P_2}[/latex]
∴ W = nRT. log[latex]\frac{P_1}{P_2}[/latex]
Equation of adiabatic process:
Consider, ‘n’ mole of gas is contained in a cylinder filled with movable piston is heated at constant volume due to which small rise in temperature is dT.
Then, according to 1st law of thermodynamics,
dQ = dU + PdV
Or, nCv.dt = dU + 0
Or, nCv.dt = dU ……………….. (i)
Now, let this gas is heated adiabatically upto same rise in temperature dT.
According to 1st law of thermodynamics,
dQ = dU + PdV
But, for adiabatic process, dQ = 0
From eqn. (1), substituting the values:
0 = nCv dt + PdV ………………………… (ii)
Now, for ‘n’ mole of gas,
PV = nRT
Differentiating both sides,
PdV + VdP = nR dT
∴ dT = [latex]\frac{PdV+VdP}{nR}[/latex]
Now, from eqn. (2), substituting the values,
0 = nCv ([latex]\frac{PdV_VdP}{nR}[/latex]) + PdV
Or, 0 = ([latex]\frac{C_vPdV+C_vVdP+PdVR}{R}[/latex])
Or, 0 = [latex](C_vPdV+C_vVdP+PdV(C_p-C_v)[/latex] [∵Cp – Cv =R]
Or, 0 = [latex](C_vPdV+C_vVdP+C_pPdV-C_vPdV)[/latex]
Or, 0 = nCvVdP + nCpPdV
Or, CvVdP = – CpPdV
Or, [latex]\frac{dP}{P}=-\frac{C_p}{C_v}\frac{dV}{V}[/latex]
Or, [latex]\frac{dP}{P} = -\gamma \frac{dV}{V}[/latex] [∵γ = [latex]\frac{C_p}{C_v}[/latex]]
Integrating both sides,
[latex]\int{\frac{dP}{P}} = -\gamma \int{\frac{dV}{V}}[/latex]
Or, log P = -γ. logV + logK
Or, log P + γ. logV = logK
Or, log P + [latex]logV^\gamma = logK[/latex]
Or, log PVγ = logK
Or, PVγ = K …………………………. (iii)
Or, P1V1γ = P2V2γ
Now, we have,
PV = RT
Or, P = [latex]\frac{RT}{V}[/latex]
Substituting this value in equation (iii),
[latex]\frac{RT}{V}V^\gamma = K[/latex]
Or, TVγ-1 = [latex]\frac{K}{R}[/latex] = K‘
We also have, PV = RT
Or, V = [latex]\frac{RT}{P}[/latex]
Substituting this value in eqn. (iii), we get,
P.[latex](\frac{RT}{P})^\gamma = K[/latex]
Or, P1 – γ . Tγ = [latex]\frac{K}{R^\gamma}=K”[/latex]
Workdone by adiabatic process:
Let us consider, n mole of an ideal gas is contained in a cylinder fitted with a movable piston with perfectly insulating walls having pressure, volume & temperature. The gas is allowed to expand adiabatically from V1 to V2. Then, workdone by the gas to expand through dV is dW = P.dV
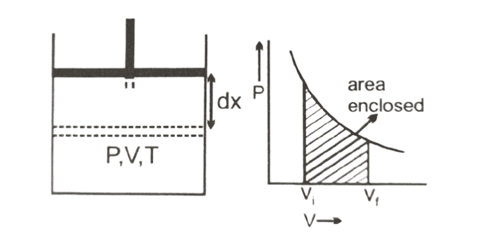
So, toal workdone (W) = [latex]\int_{V_1}^{V_2}dW = \int_{V_1}^{V_2}PdV[/latex]
For adiabatic process,
[latex]PV^\gamma = K[/latex], where, γ = [latex]\frac{C_p}{C_v}[/latex]
P = [latex]\frac{K}{V^\gamma}[/latex]
∴ W = [latex]\int_{V_1}^{V_2}\frac{K}{V^\gamma}dV[/latex]
= K[latex]\int_{V_1}^{V_2}V^{-\gamma}dV[/latex]
= K[latex][\frac{V^{-\gamma +1}}{-\gamma + 1}]_{V_1}^{V_2}[/latex] = [latex]\frac{K}{-\gamma +1}[V_2^{-\gamma + 1} – V_1^{-\gamma + 1}][/latex]
= [latex]\frac{1}{1-\gamma}[KV_2^{-\gamma + 1}-KV_1^{-\gamma + 1}][/latex]
But, P1V1γ = P2V2γ = K
∴ W = [latex]\frac{1}{1-\gamma}[P_2V_2^\gamma V_2^{-\gamma + 1} – P_1V_1^\gamma V_1^{-\gamma + 1}][/latex]
= [latex]\frac{1}{1-\gamma}[/latex][P2V2 – P1V1]
= [latex]\frac{1}{1-\gamma}[/latex][RT2 – RT1]
= [latex]\frac{R}{1-\gamma}[/latex][T2 – T1]
∴ W = [latex]\frac{R}{1-\gamma}[/latex][T2 – T1].
Since, value of [latex]\gamma[/latex] is greater than one,
W = [latex]\frac{R}{\gamma – 1}[T_1 – T_2][/latex]
This is the required expression for workdone during adiabatic process and it depends only on initial and final temperature.
Why Cp is greater than Cv?
In case of Cv, the volume is constant. So, all heat energy supplied is used to increase the internal energy of the system only. But, in case of Cp, the pressure is constant, and volume changes. So, the heat energy is required not only to increase the internal energy but also to do work against the external pressure. So, more heat will be required to increase the temperature of the gas. Hence, Cp is greater than Cv.
Limitations of First Law of Thermodynamics:
- As we know, the heat flows from a body at higher temperature to a body at lower temperature. But, the first law of thermodynamics could not indicate why the heat cannot flow from a colder to hotter body. i.e., direction of heat flow.
- It could not explain the conversion of heat energy into mechanical energy. As in, when a bullet strikes a target, the K.E. of the bullet is converted into heat energy. But 1st law could not explain the reverse conversion of heat into K.E.
- First law of thermodynamics does not give to what extent the mechanical energy is obtained from heat energy.
Finished studying this chapter resource?
Mark this chapter as complete to update your course progress.