Heat
When we touch a cup of tea, we feel hot and when we touch an ice, we feel cold. We feel hotness when our body gains some energy from another body and coldness is due to the flow of energy from our body to another body. Thus, heat is “the form of energy that flows from one body to another producing the sensation of hotness and coldness”.
When we supply heat to any object, the K.E. of the molecules of the object increases. So, total sum of K.E. of all the molecules of any object gives heat energy of that object.
Dynamic Theory of Heat
In this theory, all the substances are made of molecules and they are in random motion. The motion of molecules is of three types:
i. Translatory: It is the motion in which molecules move in a straight line. It is common in gases.
ii. Vibratory: It is the to and fro motion of the molecules about their mean position. It is common in solids and liquids.
iii. Rotatory: It is the rotation of molecules about their axes. It occurs at high temperature.
So, heat is the form of energy that remains in the body as total energy due to these motion of the molecules of the body.
Temperature
Temperature is the fundamental physical quantity which measures the degree of hotness or coldness of a body. The heat flows from higher temperature to lower temperature. In another way, temperature is the measure of average K.E. of a molecule. It can be measured in different scales like Celsius (oC), Fahrenheit (oF), Kelvin (K), Reaumer (oR) and Rankine (oRn). Among them, Kelvin (K) is the SI unit of temperature.
Unit of Heat Energy
In C.G.S. system, unit of heat energy is calorie (Cal). Whereas, the SI unit of heat energy is Joule (J). The relation between calorie and Joule is: 1 Cal = 4.18 J.
Differences between Heat and Temperature
| Heat | Temperature |
| 1. Heat is the form of energy which produces the sensation of hotness or coldness on our body. | 1. Temperature is the measure of degree of hotness or coldness of a body. |
| 2. It is the cause on a body. | 2. It is the effect on a body. |
| 3. Two bodies in thermal equilibrium, may or may not have the same amount of heat. | 3. Two bodies in thermal equilibrium, must have the same temperature. |
| 4. Heat flowing from one body to another is independent of the amount of the heat contained on them. | 4. Heat flowing from one body to another depends on the temperature. |
| 5. It is the total K.E. of all the molecules of the body. | 5. It is the average K.E. of a molecule of a body. |
Effects of Heat
The effects of heat are as follows:
i. Rise in Temperature
If heat is supplied on a body, in general, its temperature rises. However, in case of phase change (changing from solid to liquid and liquid to vapour), remains constant temperature even if heat is supplied.
ii. Rise in volume
If heat is supplied on a body, its length, breadth is increased. Thus, the volume on a body increases.
iii. Change of state of matter
If we apply heat, on a body, its state is changed. For example, if heat is applied on ice, it changes into liquid and liquid is changed into gases. So, heat changes the state of matter.
iv. Change in electric properties
If heat is supplied on a body, its temperature increases. Increase of temperature of pure metals increases the resistance of the metal.
v. Change in physical and chemical properties:
Increase of temperature on a body, causes the change in physical and chemical properties.
vi. Generation of light:
Increase of temperature, on a body, causes the generation of light.
Thermal Equilibrium
If two bodies at two different temperatures are kept in contact, heat flows from a body at higher temperature to another body at lower temperature and it continues till their temperature becomes equal and net flow of heat between them is zero. This state of equality in temperature is called thermal equilibrium.
Zeroth Law of Thermodynamics
It states that, “When two systems are separately in thermal equilibrium with a third system, then they must be in thermal equilibrium with each other.” To explain this, let us consider, three systems A, B and C in which A and B are separated by non – conducting (insulating) wall while a conducting wall separates C from A and B.
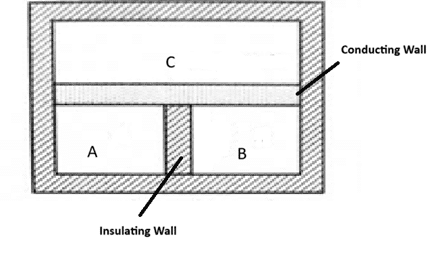
If they are let as it is, heat flows between A and C and between B and C until they attain thermal equilibrium. i.e. TA= TC and TB = TC. This implies, TA = TB.
When the insulating wall between A and B is replaced by a conducting wall, no heat flows between A and B, which shows that A and B are also in thermal equilibrium. This proves the zeroth law of thermodynamics.
Thermometry
The branch of physics, which deals with the measurement of temperature of a body is called thermometry. Thermometer is the instrument which is used to measure temperature of a body.
Different Temperature Scales
The following are the temperature scales:
a. Celsius Scale
The lower fixed point (ice point) of Celsius scale is 0oC & upper fixed point (steam point) is 100oC. The region between them is divided into 100 equal parts and each part is called 1oC.
b. Fahrenheit Scale
The lower fixed point of Fahrenheit scale is 32oF & upper fixed point is 212oF. The region between them is divided into 180 equal parts & each part is called 1oF.
c. Kelvin Scale
The lower fixed point of Kelvin scale is 273K & upper fixed point of the scale is 373K. The region between them is divided into 100 equal parts & each part is called 1K.
d. Reaumer Scale
The lower fixed point of Reaumer scale is 0oR & upper fixed point is 80oR. The region between them is divided into 80 equal parts & each part is called 1oR.
e. Rankine Scale
The lower fixed point of Rankine scale is 492oRn & upper fixed point is 672oRn. The region between them is divided into 180 equal parts & each part is called 1oRn.
Relation between different Temperature scales
Consider, five thermometers in Celsius, Fahrenheit, Kelvin, Reaumer and Rankine are kept in a warm water & the corresponding readings are C, F, K and R and Rn respectively as shown in the given figure.
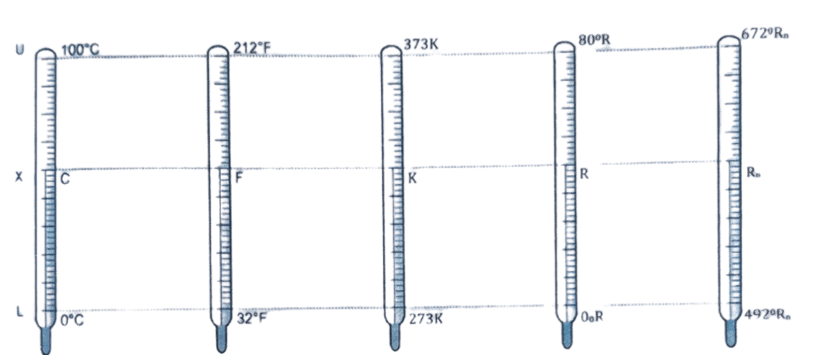
We know,
[latex]\frac{X – L}{U – L} = \frac{C-0}{100-0} = \frac{F-32}{212-32}=\frac{K-273}{373-273} = \frac{R-0}{80-0} = \frac{R_n – 492}{672-492}[/latex]
Or, [latex]\frac{C}{100} = \frac{F-32}{180} = \frac{K-273}{100} = \frac{R}{80} = \frac{R_n – 492}{180}[/latex] is the required relation.
Absolute Zero Temperature
Absolute zero is the lowest possible temperature where heat energy is absent in a substance. Absolute zero is also known as zero kelvin temperature since it measures zero on the Kelvin scale.
Note: The value of the absolute zero is -459.67 degrees Fahrenheit on the Fahrenheit scale and -273.15 degrees Celsius on the Celsius scale. The lowest temperature a thermodynamic system can withstand is -459.67 degrees Fahrenheit and -273.15 degrees Celsius. The absolute zero temperature is a theoretical value.
Finished studying this chapter resource?
Mark this chapter as complete to update your course progress.