Gas Laws
Boyle’s Law
Boyle’s law states that, “at constant temperature, the pressure exerted by a given mass of a gas is inversely proportional to the volume occupied by it.” If P be the pressure exerted by a gas of volume V, then, at constant temperature,
P ∝ [latex]\frac{1}{V}[/latex]
PV = constant
Let, P1 and V1 be the initial pressure and volume of the gas, which is changed to final pressure P2 and V2 at constant temperature T, then, Boyle’s law can be written as,
P1V1 = P2V2 = constant
This is Boyle’s law.
If we plot the graph between P & V, a hyperbolic curve is obtained. Similarly, if we plot the values of P & [latex]\frac{1}{V}[/latex], the following straight line is obtained:
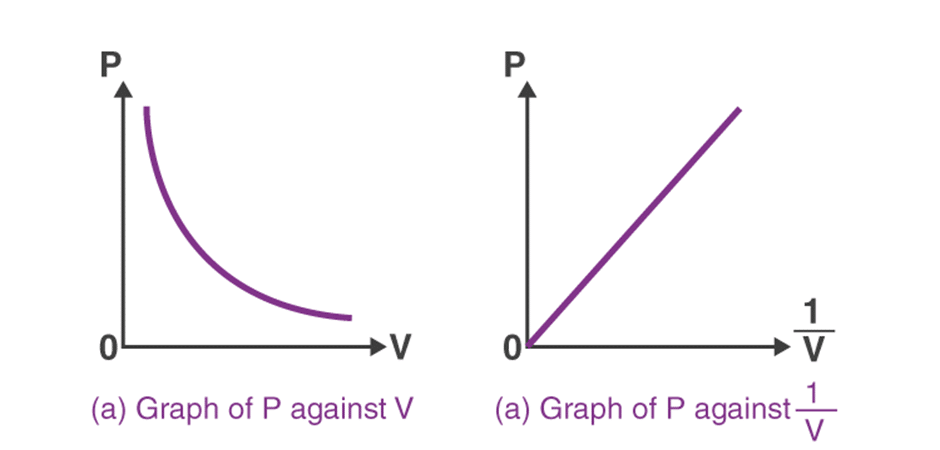
Charles’s law
a. Charles’s law at Constant Pressure (Law of volume)
It states that, “At constant pressure, the volume of the gas increases or decreases by a constant fraction of the volume at 0oC for each degree rise or fall in temperature.”
If Vo be the volume of the gas at 0oC and [latex]V_\theta^oC[/latex] be that at [latex]\theta^oC[/latex] at constant pressure, then, from Charles’s law at constant pressure,
Final volume [latex](V_\theta)[/latex] = Initial Volume (Vo) + Constant fraction [latex](\gamma_p)[/latex]. Initial volume (Vo). Change in temperature [latex](\theta)[/latex]
Or, [latex]V_\theta = V_o + \gamma_pV_o\theta[/latex], where, [latex]\gamma_p[/latex] is known as volume coefficient of gas at constant pressure.
[latex]V_\theta = V_o(1+\gamma_p\theta)[/latex]
Experimentally, [latex]\gamma_p = \frac{1}{273}[/latex]
So, [latex]V_\theta = V_o(1+\frac{1}{273}\theta)[/latex]
= [latex]V_o(\frac{273+\theta}{273}) = \frac{V_oT_\theta}{T_o}[/latex]
Or, [latex]\frac{V_\theta}{T_\theta} = \frac{V_o}{T_o}[/latex]
Or, [latex]V\propto T[/latex]. i.e., Volume of a gas is directly proportional to the absolute temperature at constant pressure, which is Charles’s law at constant pressure.
If we plot a graph between volume along y axis and temperature along x axis, a straight line is obtained, which is shown in figure alongside.
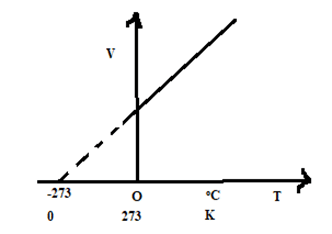
b. Charle’s law at Constant Volume (Law of Pressure)
It states that, “If volume of a given mass of a gas remains constant, the pressure of the gas increases or decreases by a constant fraction of the volume of the gas at 0oC for each degree rise or fall in temperature.”
If Po be the pressure of the gas at 0oC and [latex]P_\theta[/latex] be that at [latex]\theta^oC[/latex] at constant volume, then, from Charle’s law at constant volume,
Final pressure [latex]P_\theta[/latex] = Initial Pressure (Po) + Constant fraction [latex](\gamma_v)[/latex]. Initial pressure (Po). Change in temperature [latex](\theta)[/latex]
Or, [latex]P_\theta = P_o + \gamma_vP_o\theta[/latex], where, [latex]\gamma_v[/latex] is known as pressure coefficient of gas at constant pressure.
[latex]P_\theta = P_o(1+\gamma_v\theta)[/latex]
Experimentally, [latex]\gamma_v = \frac{1}{273}[/latex]
So, [latex]P_\theta = P_o(1+\frac{1}{273}\theta)[/latex]
[latex]P_o(\frac{273+\theta}{273}) = \frac{P_oT\theta}{T_o}[/latex]
Or, [latex]\frac{P_\theta}{T_\theta} = \frac{P_o}{T_o}[/latex]
[latex]\therefore \frac{P}{T} = Constant[/latex]
Or, [latex]P\propto T[/latex]. i.e., Pressure of a gas is directly proportional to the absolute temperature at constant volume, which is Charle’s law at constant volume.
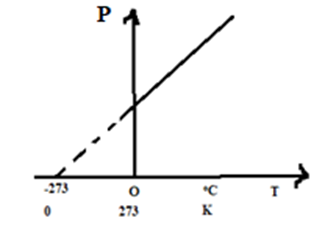
Pressure coefficient and volume coefficient
Pressure coefficient [latex](\gamma_v)[/latex]
It is defined as the increase in pressure per unit degree rise of temperature per unit original pressure at constant volume. It is denoted by [latex]\gamma_v[/latex].
Volume Coefficient [latex](\gamma_p)[/latex]
It is defined as the increase in volume per unit original volume per unit degree rise in temperature at constant pressure. It is denoted by [latex]\gamma_p[/latex].
Relation between 𝛄p and 𝛄v
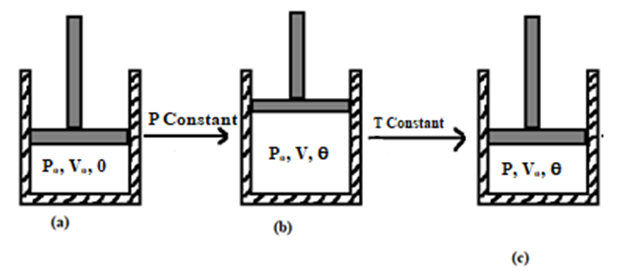
Let us consider, a certain mass of a gas is enclosed in a cylinder fitted with a frictionless piston at 0oC having initial pressure and volume to be Po and Vo respectively. Suppose the gas is heated to [latex]\theta^o C[/latex], keeping the pressure constant so that new volume becomes V, then, from Charle’s law at constant pressure,
V = [latex]V_o(1+\gamma_p\theta)[/latex] ……………. (1)
The gas is again compressed at constant temperature, so that, original volume is regained. Now, the new pressure is P. Then, according to Boyle’s law,
PoV = PVo
Or, PVo = PoV
Or, PVo = Po [Vo(1+γp [latex]\theta[/latex])]
Or, P = Po (1+γp [latex]\theta[/latex]) ……………. (2)
But, from Charle’s law at constant volume, we have,
P = Po (1+γv[latex]\theta[/latex]) …………………….. (3)
On equating (2) and (3), we have,
Po (1+γp[latex]\theta[/latex]) = Po (1+γv[latex]\theta[/latex])
Or, γp = γv
i.e., the pressure coefficient and the volume coefficient are equal.
Ideal gas equation or equation of state
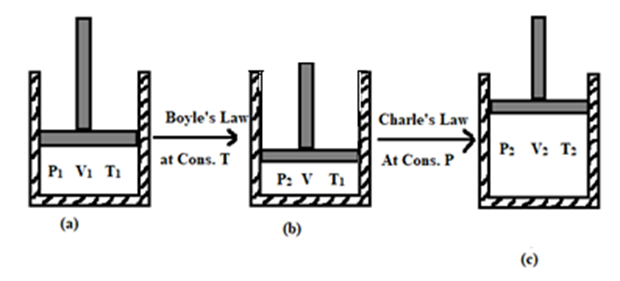
Let us consider, one mole of ideal gas is contained in a cylinder fitted with a movable piston having initial pressure P1, volume V1 & temperature T1 as shown in figure (a). Now, the gas is compressed slowly at constant temperature so that its pressure and volume changes to P2 and V. Now,
using Boyle’s law,
P1V1 = P2V
Or, [latex]V = \frac{P_1V_1}{P_2}[/latex] ………… (1)
Now, the gas is expanded at constant pressure P2 until its volume and temperature is changed to V2 and T2 respectively. Now, according to Charle’s law,
[latex]\frac{V}{T_1} = \frac{V_2}{T_2}[/latex]
[latex]\therefore V = V_2 \times \frac{T_1}{T_2}[/latex] …………….. (2)
From equations (1) and (2), we get,
[latex]\frac{P_1V_1}{P_2} = V_2\times \frac{T_1}{T_2}[/latex]
Or, [latex]\frac{P_1V_1}{T_1} = \frac{P_2V_2}{T_2}[/latex]
Or, [latex]\frac{PV}{T} = [/latex]constant
Here, the value of constant always found to be same which is called universal gas constant (R). Its value is 8.31 [latex]J mol^{-1} K^{-1}[/latex]
[latex]\therefore \frac{PV}{T} = R[/latex]
Or, PV = RT ……………………… (3)
This is the required ideal gas equation for one mole of gas.
If there are ‘n’ moles of gas, then the equation changes to:
PV = nRT …………………….. (4)
This the required ideal gas equation for n mole of gas.
Kinetic Theory of Gas
Postulates
The postulates of kinetic theory of gas are as follows:
- A gas consists of large number of small particles called molecules.
- The gas molecules are so small that the volume occupied by a single gas molecule is negligible as compared to the total volume of the vessel.
- The gas molecules are in a state of random motion. During their motion, they collide with each other and with the wall of the vessel.
- The pressure exerted by gas molecule is due to its continuous collision with each other and with the wall of the vessel.
- The collision between the gas molecules is perfectly elastic. i.e., there is no loss of Kinetic energy during collision.
- The average kinetic energy of gas molecule is directly proportional to the absolute temperature. i.e., K.E. ∝ T. where, T is absolute temperature.
(Note: the temperature measured in Kelvin scale is called absolute temperature).
Pressure exerted by a gas in a container
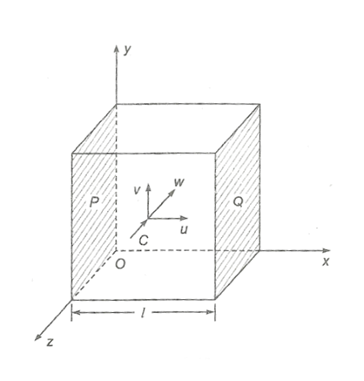
Let us consider, ‘N’ no. of molecules of a gas are contained in a cube of length ‘l’ as shown in the figure. Let, a typical molecule is moving with velocity ‘c’, whose components are u, v and w along x, y and z axis. Then, c2 = u2 + v2 + w2
Let, the molecule collide on the wall Q and bounce back with same velocity u. Then,
Its change in momentum = mu – (-mu)
= mu + mu
= 2mu
Now, the time taken by the molecule to collide on the wall Q and return back to wall P is
t = [latex]\frac{2l}{u}[/latex]
Rate of change of momentum = [latex]\frac{change\ in\ momentum}{time\ taken}[/latex]
= [latex]\frac{2mu}{2l/u} = \frac{mu^2}{l}[/latex]
So, the force exerted by the single molecule on a wall (F) = [latex]\frac{mu^2}{l}[/latex]
Now, pressure exerted (P) = F/A = [latex]\frac{\frac{mu^2}{l}}{l^2}[/latex]
= [latex]\frac{mu^2}{l^3}[/latex]
The pressure in x-axis due to all molecules,
Px = [latex]\frac{mu_1^2}{l^3} + \frac{mu_2^2}{l^3} + \frac{mu_3^2}{l^3} + …………. + \frac{mu_n^2}{l^3}[/latex]
= [latex]\frac{m}{l^3}(u_1^2 + u_2^2 + ………………. + u_n^2)[/latex] ……………. (i)
Since, mean square speed of the gas along x – axis is
[latex]\bar{u^2} = \frac{u_1^2 + u_2^2 + …………….. + u_n^2}{N}[/latex]
[latex]\bar{u^2}N = u_1^2 + u_2^2 + …………… + u_n^2[/latex] ……………… (ii)
So, from equations (i) and (ii),
The pressure (Px) = [latex]\frac{m}{l^3}\times \bar{u^2}N[/latex] ……………. (iii)
Similarly, pressure along y-axis is:
Py = [latex]\frac{m}{l^3}\times \bar{v^2}N[/latex]
And, pressure along z-axis is:
Pz = [latex]\frac{m}{l^3}\times \bar{w^2}N[/latex]
So, total average pressure (P) = [latex]\frac{P_x + P_y + P_z}{3}[/latex]
= [latex]\frac{1}{3}[\frac{m}{l^3}\times \bar{u^2}N + \frac{m}{l^3}\times \bar{v^2}N+\frac{m}{l^3}\times \bar{w^2}N][/latex]
= [latex]\frac{mN}{3l^3}[\bar{u^2}+\bar{v^2}+\bar{w^2}][/latex]
Or, P = [latex]\frac{M}{3V}\bar{c^2}[/latex]
where, [latex]\bar{c^2} = \bar{u^2} + \bar{v^2} + \bar{w^2}[/latex] is the mean square speed of gas, mN = total mass of gas (M) and volume of cube (V) = l3
So, P = [latex]\frac{1}{3}\rho \bar{c^2}[/latex] where, ρ = [latex]\frac{M}{V}[/latex]
Root Mean Square Speed (r.m.s)
The square root of the mean of the squares of the speed of gas molecules is called root mean square speed (r.m.s.).
Crms = [latex]\sqrt{\bar{c^2}}[/latex]
= [latex]\sqrt{\frac{3P}{\rho}}[/latex] [∵ P = [latex]\frac{1}{3}\rho \bar{c^2}[/latex]]
Variation of Crms with temperature and molecular mass
Crms = [latex]\sqrt{\frac{3P}{\frac{M}{V}}} = \sqrt{\frac{3PV}{M}}[/latex]
= [latex]\sqrt{\frac{3RT}{M}}[/latex] [∵PV = RT]
For a given mass of gas,
Crms ∝ [latex]\sqrt{T}[/latex]
[latex]\frac{C_1}{C_2} = \sqrt{\frac{T_1}{T_2}}[/latex]
For a given temperature,
Crms ∝ [latex]\frac{1}{\sqrt{M}}[/latex]
[latex]\frac{C_1}{C_2} = \sqrt{\frac{M_2}{M_1}}[/latex]
Average Energy with molecules of gas [latex](\bar{E_k})[/latex]
We know,
P = [latex]\frac{1}{3}\rho \bar{c^2}[/latex]
= [latex]\frac{1}{3}\frac{M}{V}\bar{c^2}[/latex]
3PV = Nm[latex]\bar{c^2}[/latex]
Or, [latex]\frac{3PV}{N} = m\bar{c^2}[/latex]
Or, [latex]\frac{3}{2}\frac{RT}{N} = \frac{1}{2}m\bar{c^2}[/latex] [∵multiplying both sides by ½]
Or, [latex]\frac{3}{2}\frac{RT}{N} = \bar{E_k}[/latex]
For 1 mole of gas
The total number of molecules is equal to Avogadro’s number. i.e., N = [latex]N_A[/latex]
[latex]\frac{3}{2}\frac{RT}{N_A} = \bar{E_k}[/latex]
Or, [latex]\frac{3}{2}KT = \bar{E_k}[/latex]
[latex]\bar{E_k} = \frac{3}{2}KT[/latex] where, K is Boltzmann Constant, K = [latex]\frac{R}{N_A}[/latex]
Derivation of gas laws
We know,
P = [latex]\frac{1}{3}\rho \bar{c^2}[/latex]
= [latex]\frac{1}{3}\frac{M}{V}\bar{c^2}[/latex]
= [latex]\frac{1}{3}\frac{M}{V}KT[/latex]
[∵ [latex]\bar{c^2} \propto T[/latex]
Or, [latex]\bar{c^2} = KT[/latex]], where K is a proportionality constant.
At constant temperature,
P ∝ 1/V, which is Boyle’s Law.
Now, at constant pressure,
V ∝ T, which is Charle’s Law at constant pressure. [∵PV = [latex]\frac{1}{3}MT[/latex]]
At constant volume,
PV = [latex]\frac{1}{3}MT[/latex]
Or, [latex]\frac{PV}{T}[/latex] = Constant
[latex]\therefore[/latex] P ∝ T, which is Charle’s Law at constant volume.
Gas Constant (R)
Gas constant (R) is defined as the amount of workdone per mole per unit kelvin. We know,
PV = nRT
[latex]\therefore R = \frac{PV}{nT}[/latex]
[latex]= \frac{Nm^{-2}m^3}{mol.K} = \frac{N.m}{mol.K} = Jmol^{-1}K^{-1}[/latex]
It is a constant quantity. i.e.
R = 8.31 [latex]J\ mol^{-1}K^{-1}[/latex] (In SI)
R = 1.98 [latex]Cal\ mol^{-1}[/latex]o[latex]C^{-1}[/latex] (In CGS)
Derivation of ideal gas equation from kinetic theory of gas
From the kinetic theory of gas, the pressure exerted by gas on the wall of container is:
P = [latex]\frac{1}{3}\rho \bar{c^2} = \frac{1}{3}\frac{M}{V}\bar{c^2}[/latex]
Or, P = [latex]\frac{Mc^2}{3V}[/latex] ……………. (3)
And also,
C = [latex]\frac{3RT}{M}[/latex] …………. (4)
From equation (3) & (4), we get,
P = [latex]\frac{1}{3V}M.(\sqrt{\frac{3RT}{M}})^2[/latex]
= [latex]\frac{1}{3V}M.\frac{3RT}{M} = \frac{RT}{V}[/latex]
[latex]\therefore[/latex] PV = RT ……………… (5)
This proves the ideal gas equation for one mole of gas.
Finished studying this chapter resource?
Mark this chapter as complete to update your course progress.