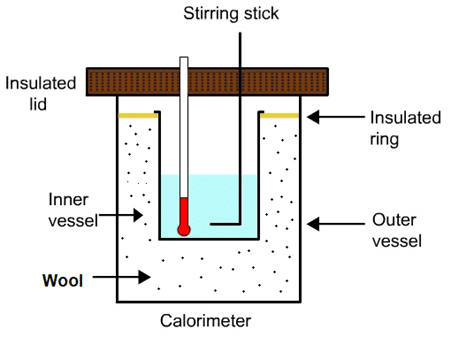
Calorimeter
The vessel in which heat exchange in between different bodies at different temperature is observed is called calorimeter. It is made of copper with copper stirrer kept inside a wooden box having lid. A thermometer can be inserted from the hole on the liq. A woollen cloth is kept into the gap between vessel and wooden box.
Heat Equation and Specific Heat Capacity
When same amount of heat is given to a substance of same material but having different masses, the rise in their temperature is also found to be different. Experimentally, it has been found that, the amount of heat (Q):
- required to raise the temperature of substance by certain value [latex](\Delta \theta)[/latex] is directly proportional to the mass (m) of the substance. i.e.
[latex]Q \propto m[/latex] ………… (i) - required for the same mass of the substance is directly proportional to the rise in temperature [latex](\Delta \theta)[/latex]. i.e.
[latex]Q \propto \Delta \theta[/latex] ………. (ii)
Combining equations (i) and (ii), we get,
[latex]Q \propto m(\Delta \theta)[/latex]
[latex]\therefore Q = ms\Delta \theta[/latex] ………….. (iii)
This equation is called the heat equation and the proportionality constant ‘s’ is called the specific heat capacity of the body. From equation (iii), we have,
[latex]s = \frac{Q}{m\times \Delta \theta}[/latex]
When m = 1 unit and [latex]\Delta \theta[/latex] = 1 unit, then [latex]s = Q[/latex] Thus, the specific heat capacity of a body is defined as the amount of heat required to raise the temperature of unit mass of a body through unit change in temperature.
Unit of Specific Heat Capacity
In SI system, its unit is: JKg-1K-1 and in CGS system it is measured in Calg-1K-1.
Heat capacity or Thermal Capacity
The amount of heat required to raise the temperature of whole mass of a body through unit temperature difference is called thermal capacity of the body.
If Q be the amount of heat required to raise the temperature of m mass of a body through 1oC or 1K, then, from heat equation,
Or, Q = [latex]ms\times 1[/latex]
Or, [latex]Q = ms[/latex]
Or, Q = c, where, c = ms = heat capacity = product of mass and specific heat capacity.
Its unit is [latex]JK^{-1}[/latex] in SI and [latex]Cal ^oC^{-1}[/latex] in CGS.
Water Equivalent of a Substance
When a liquid is heated, some of the heat supplied is absorbed by the vessel containing liquid. So, water equivalent of the substance is defined as the amount of water which absorbs the same amount of heat as done by the substance for the same rise or fall of temperature.
Let, Q be the amount of heat absorbed by a substance of mass ‘m’ and specific heat ‘s’ to rise its temperature through , then, from heat equation,
Q = [latex]ms\Delta \theta[/latex] ………. (i)
Again, if ‘w’ be the amount of water that absorbed same amount of heat for same rise in temperature, then,
[latex]Q = w.s_w\Delta \theta[/latex]……… (ii), where [latex]s_w[/latex] be the specific heat capacity of water.
From equations (i) and (ii),
[latex]ms\Delta \theta = w.s_w\Delta \theta[/latex]
Or, [latex]w.s_w = ms[/latex]
[latex]\therefore w = ms[/latex] in CGS system. [latex][\because s_w = 1\ in\ CGS\ System][/latex]
Principle of Calorimetry
When two bodies at different temperatures are kept in thermal contact, the body at higher temperature loses heat while the body at lower temperature gains heat. So, principle of calorimetry states that, “if there is no exchange of heat with the surroundings, heat lost by hot body is always equal to heat gained by cold body.” i.e., heat lost = heat gained.
Determination of Specific Heat Capacity of Solid by Method of Mixture
To determine specific heat capacity of solid, a small ball of it is weighed and heated in a steam chamber where, ball is suspended with the help of string until it gets steady temperature. Meanwhile, a dried cleaned calorimeter is weighed. Then, calorimeter is filled with water upto 2/3 of its volume & kept in wooden box.
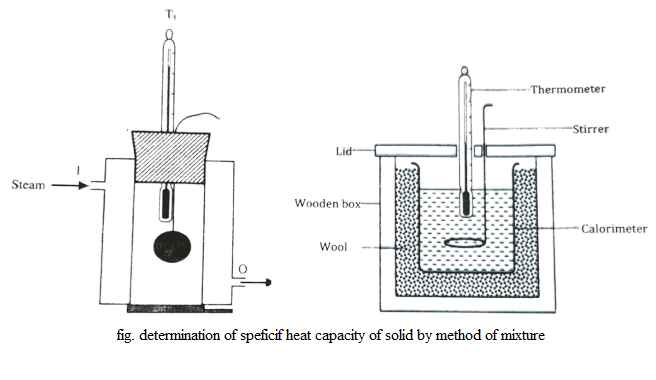
Also, the temperature of water is noted. When the temperature of solid becomes steady, the calorimeter is carried below the hole of steam chamber & the ball is dropped inside the calorimeter from the chamber. Then, by covering with lid, the calorimeter is returned back. By stirring the mixture, constant temperature is noted.
Let mass of solid = m
Mass of calorimeter with stirrer = mc
Mass of water = mw
Specific heat capacity of solid = s
Specific heat capacity of material of calorimeter = Sc
Specific heat capacity of water = Sw
Steady temperature of solid = [latex]\theta_1[/latex]
Initial temperature of water = [latex]\theta_2[/latex]
Final temperature of mixture = [latex]\theta[/latex]
Heat lost by solid = ms[latex](\theta_1 – \theta)[/latex]
Heat gained by calorimeter & water = mCSC [latex](\theta – \theta_2)[/latex] + mwSw[latex](\theta – \theta_2)[/latex]
= (mCSC + mwSw) [latex](\theta – \theta_2)[/latex]
By the principle of calorimetry,
ms[latex](\theta_1 – \theta)[/latex] = (mCSC + mwSw) [latex](\theta – \theta_2)[/latex]
[latex]\therefore s = \frac{(m_cS_c+m_wS_w)(\theta – \theta_2)}{m(\theta_1 – \theta)}[/latex]
By knowing all the terms of RHS, the specific heat capacity of solid can be determined.
Newton’s law of cooling
Statement
It states that, ‘the rate of cooling (i.e., decrease of heat) is directly proportional to the difference in temperature of body & its surrounding.’
If Q be the heat lost by hot body at temperature [latex]\theta[/latex] in time dt, then, by Newton’s law of cooling,
[latex]\frac{dQ}{dt}\propto (\theta – \theta_s)[/latex]
Where, [latex]\theta_s[/latex] = temperature of surrounding
[latex]-\frac{dQ}{dt} = K(\theta – \theta_s)[/latex]
[latex]\frac{dQ}{dt} = – K(\theta – \theta_s)[/latex] ……………………………… (1)
Where, negative sign shows that, as ‘t’ increases, Q decreases & K is the proportionality constant.
The value of K depends upon:
- The nature of the substance
- Surface area of surface expressed to surrounding
If ‘m’ is the mass & ‘s’ is the specific heat capacity of the body, the heat lost in time dt by the body to the surrounding and fall in temperature of body is [latex]\theta[/latex], then,
[latex]\frac{dQ}{dt} = ms\frac{d\theta}{dt}[/latex] …………………….. (2)
From eqn. (1) & (2), we have,
[latex]ms\frac{d\theta}{dt} = -K(\theta – \theta_s)[/latex]
Or, [latex]\frac{d\theta}{\theta – \theta_s} = \frac{-K}{ms}dt[/latex]
Integrating both sides, we get,
[latex]\int{\frac{d\theta}{\theta – \theta_s}} = – \int{\frac{K}{ms}dt}[/latex]
Or, [latex]log(\theta – \theta_s) = -\frac{K}{ms}t + C[/latex], where, C is integration constant.
[latex]\therefore log(\theta – \theta_s) = -\frac{K}{ms}t + C[/latex]
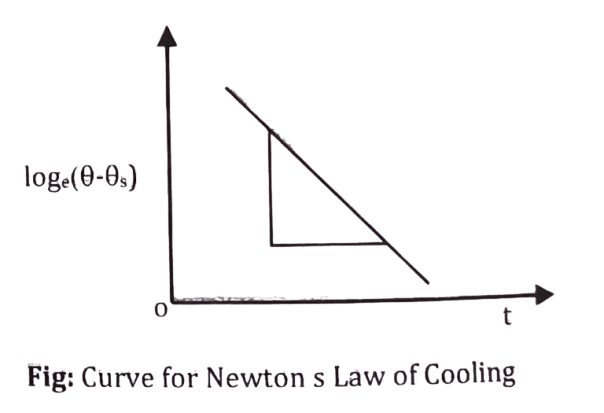
This equation is the equation of straight line which verifies the Newton’s law of cooling.
Determining specific heat capacity of liquid by method of cooling
Newton’s law of cooling can be used to determine the specific heat capacity of the liquid by the following procedure:
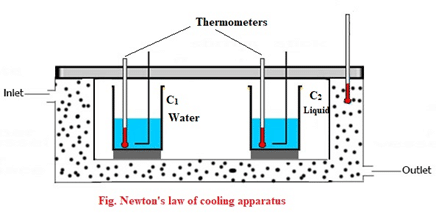
- The Calorimeter C1 & C2 are weighed.
- Some water in a beaker and same volume of liquid in another beaker is taken and both are heated upto temperature [latex]\theta_2[/latex].
- Then water and liquid are taken in calorimeters C1 and C2.
- They are placed inside a cooling apparatus.
- The cooling apparatus maintains constant temperature inside due to continuous flow of oil.
- Now, temperatures of both water and liquid are noted after regular interval of time.
- A graph of temperature against time is plotted, which is shown in figure below. Then, time taken by water and liquid to cool through same range of temperature are obtained. Finally, both calorimeters are weighted again to know the mass of water and liquid.
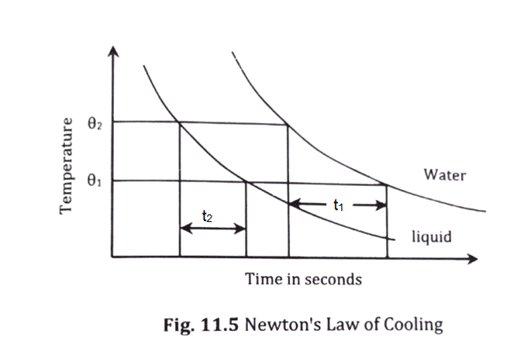
Let the specific heat capacity of the calorimeter = Sc
Mass of calorimeter C1 = m1
Mass of calorimeter C2 = m2
Mass of water in C1 = mw
Mass of equal vol. of liquid in C2 = ml
The specific heat capacity of the liquid = S
The specific heat of water = Sw
Time taken by water to cool from [latex]\theta_2[/latex] to [latex]\theta_1[/latex] = t1 &
Time taken by the liquid to cool from [latex]\theta_2[/latex] to [latex]\theta_1[/latex] = t2
The rate of loss of heat by water & calorimeter C1
[latex](\frac{dQ}{dt})_1 = \frac{(m_wS_w + m_1S_c)(\theta_2 – \theta_1)}{t_1}[/latex]
& the rate of loss of heat by liquid & calorimeter C2
[latex](\frac{dQ}{dt})_2 = \frac{(m_lS + m_2S_c)(\theta_2 – \theta_1)}{t_2}[/latex]
Since, by Newton’s law of cooling, the rate of loss of heat by the two liquids must be same.
We have,
[latex]\frac{(m_wS_w + m_1S_c)(\theta_2 – \theta_1)}{t_1} = \frac{(m_lS + m_2S_c)(\theta_2 – \theta_1)}{t_2}[/latex]
[latex]\therefore (m_lS + m_2S_c) = (m_wS_w + m_1S_c)\frac{t_2}{t_1}[/latex]
[latex]\therefore S = \frac{(m_wS_w + m_1S_c)t_2}{m_lt_1} – \frac{m_2S_c}{m_l}[/latex]
Substituting the values of all known quantities, the specific heat capacity of liquid can be determined.
Change of State
We know that, the matter exists in three states – solid, liquid & gas. As temperature increases, solid changes to liquid & liquid changes to gas. The temperature at which the matter changes its state from solid to liquid is called melting point. As the liquid is further heated, it changes into vapor state. The temperature at which the matter changes its state from liquid to gas is called boiling point.
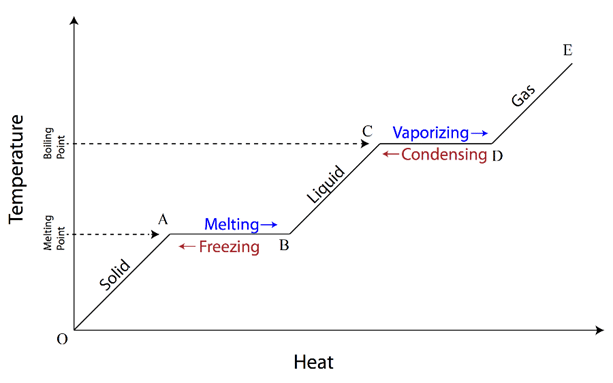
The phase change of a substance from solid to vapour (gas) is shown in graph alongside. Here, at point O, the substance is in solid state. On heating the substance, its temperature rises (represented by line OA). Point A corresponds the start of conversion of solid into liquid at melting point. At point B, all solid is converted into liquid. If we further supply heat to the liquid, the temperature rises until it reaches to point C. Here boiling starts and it begins to convert liquid to vapour state (represented by line CD). At point D, all liquid is converted into vapour. If further heat is supplied, the temperature of vapour rises.
Latent Heat
The amount of heat required to convert a substance from one phase to another at a constant temperature is called latent heat. It has been found that the amount of heat required to change the state of a substance at constant temperature is directly proportional to mass of the body. Let Q be the total quantity of heat required to change the state of m mass at constant temperature. Then,
[latex]Q \propto m[/latex]
Or, Q = Lm
Or, Q = mL; where, L is proportionality constant called specific latent heat of the body.
The latent heat is of two types:
- Latent heat of fusion: It is defined as the amount of heat required to convert unit mass of substance from its solid state to liquid state at its melting point. It is denoted by Lf. Its unit is: J/kg in SI system and cal/g in CGS system.
- Latent heat of Vaporization: It is defined as the amount of heat required to convert unit mass of substance from its liquid state to vapour state at its boiling point. It is denoted by Lv. Its unit is: J/kg in SI system and cal/g in CGS system.
Latent heat of Fusion of Ice by the method of mixture
It is defined as the amount of heat required to change unit mass of ice to water at 0oC at the same temperature.
Determination of latent heat of fusion of ice by method of mixture
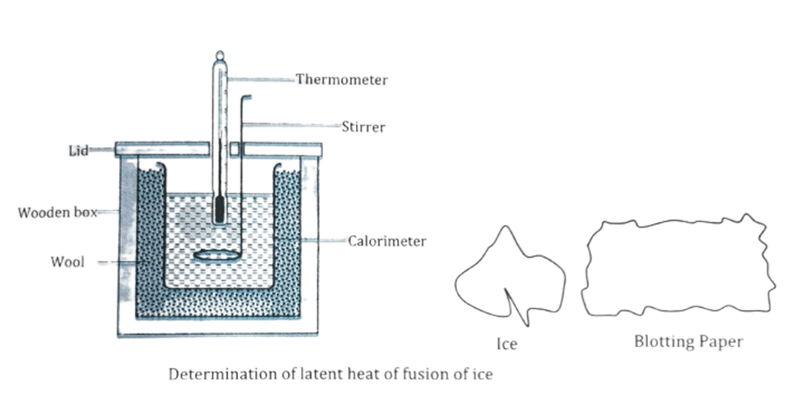
- The mass of calorimeter with stirrer is weighed.
- Water is poured into the calorimeter upto 2/3rd part of its volume & its weight is taken.
- Temperature of water in the calorimeter is noted.
- Some ice is dropped into the calorimeter after making them dry with the help of blotting paper.
- The mixture of water & ice is stirred until all ice melts & temperature of mixture is noted.
- The calorimeter is again weighted to know the mass of ice melted.
Calculation
Mass of calorimeter with stirrer = mc
Mass of water = mw
Mass of ice = mi
Specific heat capacity of calorimeter = sc
Specific heat capacity of water = sw
Latent heat of fusion of ice = Lf (unknown)
Let, initial temperature of water and calorimeter = [latex]\theta_1[/latex]
Initial temperature of melting ice = 0oC
Final temperature of mixture = [latex]\theta[/latex]
Heat lost by water & calorimeter = (mwSw + mcSc)([latex]\theta_1 – \theta[/latex])
Heat gained by ice at 0oC in melting and rising the temperature to [latex]\theta^oC[/latex]
= [latex]m_iL_f + m_is_w(\theta – 0)[/latex]
= [latex]m_iL_f + m_is_w\theta[/latex]
Now, by the principle of calorimetry,
Heat lost = Heat gained
(mwsw + mcsc)([latex]\theta_1 – \theta[/latex]) = [latex]m_iL_f+m_is_w\theta[/latex]
[latex]m_iL_f = (m_ws_w + m_cs_c)(\theta_1 – \theta) – m_is_w\theta[/latex]
[latex]\therefore L_f = \frac{(m_ws_w+m_cs_c)(\theta_1-\theta)}{m_i}-s_w\theta[/latex]
By knowing the values of RHS, the latent heat of fusion of ice is determined.
Latent heat of vaporization of water
It is defined as the amount of heat required to change unit mass of water at 1000C into vapour at same temperature.
Determination of the latent heat of vaporization of steam by the method of mixture
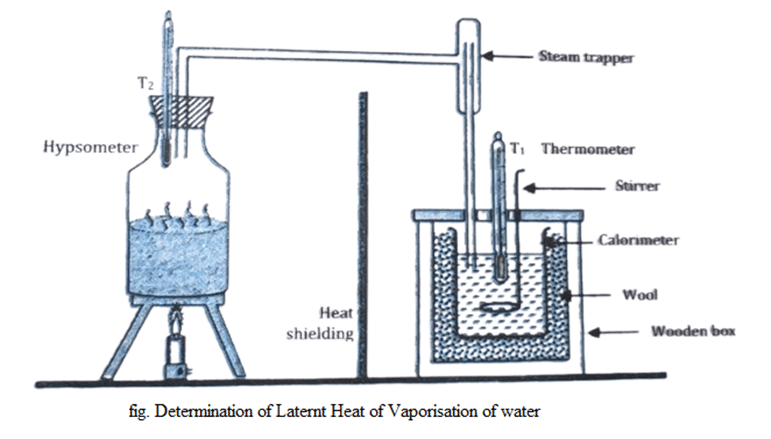
- The mass of calorimeter with stirrer is taken.
- About 2/3rd part of calorimeter is filled with water and its mass is measured & along with initial temperature of water and calorimeter.
- Steam is generated in a steam generator & passed into the water in the calorimeter. Temperature of the steam is noted.
- After some time, the steam generator is turned off and the temperature of mixture is noted.
- Once again, the calorimeter is weighted to know the mass of steam passed into the system.
Calculation
Let, mass of calorimeter with stirrer = mc
Mass of water = mw
Mass of steam = ms
Initial temperature of water and calorimeter = [latex]\theta_1[/latex]
Initial temperature of steam = 100oC
Final temperature of mixture = [latex]\theta[/latex]
Specific heat of calorimeter = sc
Specific heat of water = sw
Latent heat of steam = Lv
Total heat lost by steam upto lowering its temperature from 100oC to [latex]\theta[/latex]
= msLv + mssw(100 – [latex]\theta[/latex])
Total heat gained by water and calorimeter = mwsw([latex]\theta – \theta_1[/latex]) + mcsc([latex]\theta – \theta_1[/latex])
= ([latex]\theta – \theta_1[/latex]) (mwsw + mcsc)
By principle of calorimetry,
Heat lost = Heat gained
msLv + mssw(100 – [latex]\theta[/latex]) = ([latex]\theta – \theta_1[/latex]) (mwsw + mcsc)
Or, msLv = ([latex]\theta – \theta_1[/latex]) (mwsw + mcsc) – mssw(100 – [latex]\theta[/latex])
[latex]\therefore[/latex] Lv = [latex]\frac{(m_ws_w + m_cs_c)(\theta – \theta_1)}{m_s} – s_w(100-\theta)[/latex]
By knowing the values of RHS, the latent heat of vaporization of water is determined.
Effect of Pressure on Melting Point
During the phase change of solid into liquid, its volume changes. Most metals like copper, gold, etc. expands on melting while few solids like Rubber, ice contracts on melting. The substance which expands on melting, its molecule need energy to do work against intermolecular force and against atmospheric pressure. So, when atmospheric pressure is increased, the molecules need more energy to overcome this than previously. Hence, its melting point is increased.
Similarly, the substance which contracts on melting, on applying pressure helps the molecules go closer which helps in contraction. So, its melting point decreases.
Effect of Pressure on Boiling Point
When liquid boils, it is converted into vapor and its volume increases. To increase the volume, energy is required to do work against intermolecular force and external pressure. So, when pressure is increased, molecules need more energy to overcome it. Hence, boiling point is increased.
Evaporation and Boiling
The slow and silent conversion of the liquid from its liquid to gaseous state is called evaporation. This occurs only at the surface of the liquid. It can take place at any temperature. During this process, cooling of liquid is observed because the molecules carrying high kinetic energy escapes from the liquid surface.
The process in which liquid is converted into vapour without changing its temperature is called boiling. It occurs throughout the volume of the liquid. It takes place only at boiling point.
Phase Indicator Diagram
- Melting Line: It represents the points where solid and liquid phases exists in equilibrium.
- Vaporization Line: It represents the points where liquid and vapour phases are in equilibrium.
- Sublimation Line: The process by which a solid change directly into the vapour phase without passing through the liquid phase is called sublimation. Figure shows melting, vaporization and sublimation line.
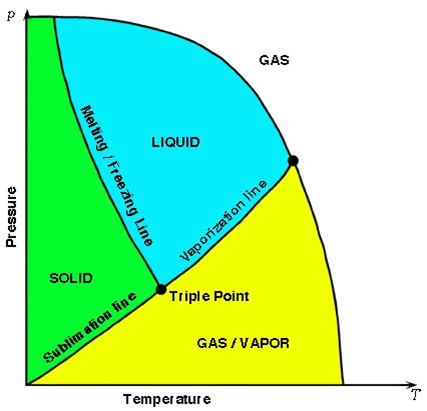
Triple Point
It is defined as a point in a phase diagram at particular pressure and temperature in which solid, liquid and gaseous state of the substance co-exists. For water, the triple point occurs at 0.01oC and 4.58 mm of Hg pressure.