Introduction
There are 3 modes by which heat is transferred:
i. Conduction
ii. Convection and
iii. Radiation
i. Conduction
The transfer of heat within a medium from one point to another without actual movement of molecules is called conduction. It is carried by means of collision between the vibrating molecules. It is applicable in solid only.
Thermal Conductivity (K)
The measure of ability of solid to conduct heat through it is called thermal conductivity. Let us consider a solid rod of length [latex](l)[/latex] and cross-sectional area (A), whose one face is maintained at higher temperature [latex]\theta_1[/latex] and other face is maintained at lower temperature [latex]\theta_2[/latex].
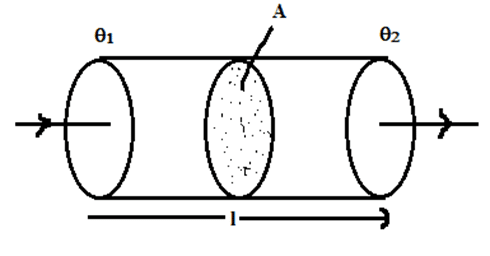
It has been found that, the quantity of heat (Q) flowing through the rod is:
- Directly proportional to the cross-sectional area. i.e., Q ∝ A
- Directly proportional to the temperature difference between the ends of the conductor. i.e., Q ∝ [latex](\theta_1 – \theta_2)[/latex]
- Inversely proportional to the separation between the two faces. i.e., [latex]Q\propto \frac{1}{l}[/latex]
- Directly proportional to the time for which heat flows. i.e., Q ∝ t
Combining all, we get,
Q ∝ [latex]\frac{A(\theta_1 – \theta_2)t}{l}[/latex]
Q = K [latex]\frac{A(\theta_1 – \theta_2)t}{l}[/latex], where, K is proportionality constant, called thermal conductivity.
When, A = 1 m2, [latex]\theta_1 – \theta_2[/latex] = 1 K, t = 1 sec and [latex]l[/latex] = 1 unit, then,
K = Q
Therefore, the thermal conductivity of solid is defined as the amount of heat flowing through a conductor having unit cross-sectional area of unit length maintained at unit temperature difference between the end of the conductor in one second.
Again, we have, Q = [latex]\frac{KA(\theta_1 – \theta_2)t}{l}[/latex]
Since, [latex]\frac{(\theta_1 – \theta_2)}{l} = \frac{d\theta}{l}[/latex] = Temperature gradient.
Q = K A [latex]\frac{d\theta}{l}t[/latex]
[latex]\frac{Q}{t} = KA\frac{d\theta}{l}[/latex]
[latex]\therefore K = \frac{Q/t}{A(\frac{d\theta}{l})}[/latex]
Again, thermal conductivity of the conductor is the rate of flow of heat per unit cross-sectional area per unit temperature gradient.
Temperature Gradient
The fall of temperature with distance between two ends of the rod in direction of flow of heat is called temperature gradient.
Unit of thermal conductivity
We know,
K = [latex]\frac{\frac{Q}{t}}{A(\frac{d\theta}{l})}[/latex]
= [latex]\frac{J/s}{m^2K/m} = (Js^{-1})m^{-1}K^{-1}[/latex]
= Wm-1K-1 [∵ W = Js-1]
Thermal Conductivity of a Good Conductor by Searle’s Method
A simple construction of Searle’s apparatus is shown in the figure below. The bar AB is taken whose thermal conductivity is to be measured, with cavities C1 & C2, the end A is connected to a steam chamber to heat it up & the end B is wounded with a copper tube in which cold water is flowing. It is surrounded by an insulator to avoid heat radiation. The thermometers T1 & T2 measures temperatures [latex]\theta_1[/latex] & [latex]\theta_2[/latex] respectively. Small amount of mercury is also poured in the cavities C1 & C2 so that thermometer responds quickly.
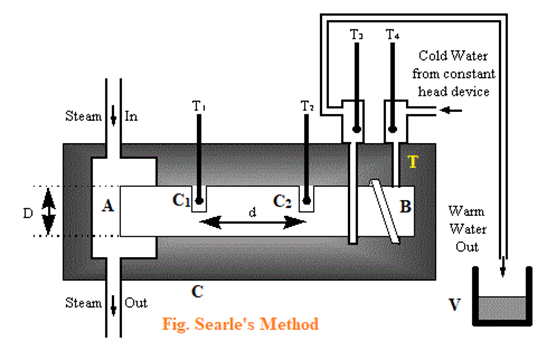
The thermometers T4 & T3 measures the temperatures of inlet & outlet of water respectively. The water coming from outlet is collected in a vessel (V).
When the steam generator is set into operation, the end A of the metal bar begins to be heated. The heat is conducted towards the end B. The temperatures in thermometers begin to rise. After certain time, a steady state is reached where all thermometers show constant readings.
If A is the area of cross-section of metal rod AB, the heat that has flown through the section between the cavities C1 & C2 in the time ‘t’ second is given by:
Q1 = [latex]\frac{KA(\theta_1 – \theta_2)t}{d}[/latex] ………………………………. (i)
If ‘m’ is the mass of water collected in the vessel (V) in time (t) and ‘s’ is the specific heat of water, the heat absorbed by the water is given by:
Q2 = [latex]m.s.(\theta_3 -\theta_4)[/latex] …………………………… (ii)
For steady state, Q1 = Q2
[latex]\therefore \frac{KA(\theta_1 – \theta_2)t}{d} = m.s.(\theta_3 – \theta_4)[/latex]
Or, [latex]K = \frac{ms(\theta_3 – \theta_4)}d{A(\theta_1 – \theta_2)t}[/latex]
By knowing all the values of RHS, the thermal conductivity (K) of good conductor is determined.
Application of Conduction
- Cooking utensils are made with metal like iron, copper, etc but provided with handles of bad conductors. Since, metals are good conductors of heat, which is easily conducted through the base and handles are bad conductors so there is no conduction of heat to our hand.
- Woollen clothes are preferred in winder because it is bad conductor of heat which prevents heat flow from our body to surrounding.
- Metal knob of door is colder than wooden parts in winter because metal knob is good conductor of heat. So, our body easily lost heat to metal knob, when touched.
ii. Convection
The process of transfer of heat within a medium from one point to another due to actual movement of the molecules is called convection. This phenomenon is mainly applicable in liquid only.
Convective Coefficient
Experimentally, it has been found that, the rate of transfer of heat [latex](\frac{Q}{t})[/latex] is
- Directly proportional to the surface area of the fluid exposed to surrounding (A). i.e.,
[latex]\frac{Q}{t}\propto A[/latex] - Directly proportional to the temperature difference between two parts of the fluid [latex](\Delta \theta)[/latex]. i.e.,
[latex]\frac{Q}{t}\propto \Delta \theta[/latex]
Combining both, we get,
[latex]\frac{Q}{t}\propto A\Delta\theta[/latex]
Or, [latex]\frac{Q}{t} = \beta A\Delta\theta[/latex], where, [latex]\beta[/latex] is known as convective coefficient
Here,
[latex]\beta = \frac{\frac{Q}{t}}{A\Delta\theta}[/latex]
If A = 1 m2, [latex]\Delta \theta = 1\ K[/latex] then [latex]\beta = \frac{Q}{t}[/latex]
Hence, convective coefficient is defined as the rate of flow of heat through the fluid having unit surface area and unit temperature difference between two parts of the fluid.
Application of Convection
- It is applicable in ventilation. The rooms are provided with ventilators. Air inside room becomes warmer and its density becomes low. It rises up and fresh cold air enters from outside.
- Trade winds are formed due to convection. The equator region of earth is heated by sun more than poles. So, air from equator moves up towards the pole. Due to this cold air from pole moves towards the equator forming wind, called trade winds.
iii. Radiation
The process of transfer of heat from one place to another with the speed of light without heating the intervening medium is called radiation. Medium is not required during radiation because the rapidly moving molecules of hot body emit electromagnetic waves with travels with velocity of light.
Reflection, Absorption and Transmission of Heat Radiation
When a heat radiation falls upon surface of any object, part of it is reflected, part of it is absorbed and part of it is transmitted out.
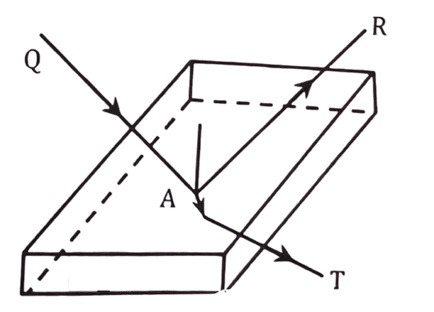
Let us consider, a heat radiation of energy Q falls on surface of any object. ‘R’ amount of heat reflects, ‘A’ amount of heat is absorbed and ‘T’ amount of heat is transmitted out. So, mathematically, we can write,
Q = R + A + T
Dividing both sides by Q, we get,
1 = [latex]\frac{R}{Q} + \frac{A}{Q} + \frac{T}{Q}[/latex]
Or, 1 = r + a + t
where, r = [latex]\frac{R}{Q}[/latex] = reflectance, a = [latex]\frac{A}{Q}[/latex] = absorbance and t = [latex]\frac{T}{Q}[/latex] = transmittance.
Black Body
The body which absorbs most of the radiations fallen upon it called Black body. Similarly, the body which absorbs all the radiation fallen upon it is called perfectly black body. When a black body is heated at suitable temperature, it can emit radiation captured by it. Thus, a black body is called good absorber as well as good emitter.
Ferry’s Black Body
Ferry devised a perfect black body. The principle is that the space is capable of absorbing all the rays falling in it.
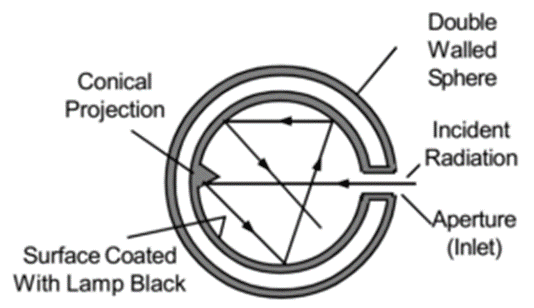
A simple construction is shown in the given figure. It consists of a double walled metallic sphere having small opening, opposite to a conical projection. The inner wall of the sphere is polished with lamp black. A heat coil is placed between the two walls for heating at different temperatures with electric current. Any radiations entering the hollow sphere through small opening is reflected many times inside the sphere. The black lamp & the conical projections trap the radiations. As a result, almost entire radiation is absorbed. When this black body is heated, the radiation will come out from the hole. So, the opening acts as a black body radiator.
Stefan’s Boltzmann’s Law of Black Body Radiation
When a black body is heated to suitable temperature, it can emit radiation captured by it. Stefan’s Boltzmann law states that, “The energy radiated per unit area per second by a perfectly black body is directly proportional to the fourth power of absolute temperature of the body.” i.e.
E ∝ T4, where, E is energy radiated per unit area per second.
E = [latex]\sigma[/latex]T4 …….. (i)
where, [latex]\sigma[/latex] is proportionality constant, called Stefan’s constant. Its value is [latex]5.67\times 10^{-8}[/latex] Wm-2K4.
So, rate of heat energy radiated (P) = [latex]\sigma AT^4[/latex] ………… (ii) where, A = area of perfect black body.
If the body is not perfectly black body, then
P = [latex]e\sigma AT^4[/latex] ………….. (iii)
where, ‘e’ be emissivity of the body, whose value ranges from 0 to 1
If the black body is enclosed by an enclosure at temperature (To), then,
P = [latex]Ae\sigma (T^4 – T_o^4)[/latex] ………… (iv)
Application of Heat Radiation
- White cloths are used in summer because they are good reflector of heat and reflects most of the heat incident on it. Dark cloths are used in winter because they are good absorber and absorbs most of heat.
- Polished and shining reflectors are used in electric heaters to reflect maximum heat in room.
- The bottom of cooking utensils is black and rough, which are good absorbers and good emitters. So, it absorbs more heat and food is cooked easily.
Solar Constant
The amount of heat radiation received from the sun per unit area per second by a perfectly black body placed on earth perpendicular to direction of rays is called solar constant (S), which is approximately equal to 1400 W/m2
i.e., [latex]S = \frac{E}{A.t} = \frac{P}{A}[/latex]
Or, P = A.S
Estimation of Surface Temperature of Sun
Let us consider, the sun of radius ‘R’ and solar constant ‘S’ is a perfectly black body at absolute temperature T. Let, r be the distance of earth from the sun.
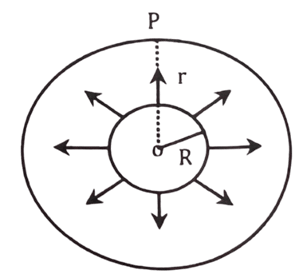
Now, from Stefan’s law, total energy radiated per unit area per second by the sun is,
[latex]E = \sigma T^4[/latex]
Now, energy radiated per unit time is: [latex]P = \sigma AT^4[/latex]
= [latex]\sigma 4\pi R^2T^4[/latex] ……….. (i) where, A = surface area of sun
Again, total energy received per second by the sphere is
[latex]P = 4\pi r^2.S[/latex] …………….. (ii), where, S = solar constant
From equations (i) and (ii), we get,
[latex]\sigma 4\pi R^2T^4 = S.4\pi r^2[/latex]
Or, T= [latex](\frac{r^2S}{R^2\sigma})^{1/4}[/latex]
Here, [latex]S = 1400\ Wm^{-2}[/latex]
R = [latex]7\times 10^8[/latex] m
r = [latex]1.5\times 10^{11}[/latex] m
[latex]\sigma = 5.67\times 10^{-8}\ Wm^{-2}K^{-4}[/latex]
Then, T = 5802.73 K [latex]\approx[/latex] 6000 K
Hence, the temperature of the sun is approximately 6000 K.
Finished studying this chapter resource?
Mark this chapter as complete to update your course progress.