The increase in size of a body on heating is called thermal expansion.
Coefficients of Expansion
When a body is heated, it expands in all dimensions i.e., increase in length, breadth and thickness simultaneously. Thus, the thermal expansion is of three types: linear expansion, superficial expansion and cubical expansion. Experimentally, it is found that increase in dimension is directly proportional to the original dimension and temperature.
- Linear Expansion
If a substance in the form of rod or wire is heated, its length increases. Such type of expansion is called linear expansion. It does not mean that there is no expansion in area and volume. However, the expansion of area and volume in insignificant as compared to the expansion in length.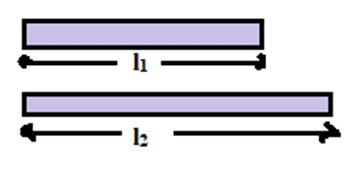
Let a metal rod has length l1 at temperature q1. When it is heated up to the temperature q2, its length increases & becomes l2. Thus, the change in length [latex](\Delta l) = l_2-l_1[/latex] & change in temperature [latex](\Delta \theta) = \theta_2 – \theta_1[/latex]. Thus, the change in length [latex](\Delta l)[/latex] is directly proportional to
(i) The original length, i.e.
[latex]\Delta l \propto[/latex] l1
The rise in temperature, i.e.
[latex]\Delta l \propto \Delta \theta = (\theta_2 – \theta_1)[/latex]
Combining both, we get,
[latex]\Delta l \propto l_1 (\theta_2 – \theta_1)[/latex]
[latex]\Delta l = \alpha l_1(\theta_2 – \theta_1)[/latex] ……….. (1)
Where, [latex]\alpha[/latex] is a proportionality constant, called coefficient of linear expansion.
From equation (1), we get,
[latex]\alpha = \frac{\Delta l}{l_1 (\theta_2 – \theta_1})[/latex] ………………………………….. (2)
= [latex]\frac{change\ in\ length}{original\ length \times change\ in\ temperature}[/latex]
If l1 = 1 unit, [latex]\theta_2 – \theta_1[/latex] = 1 unit, then, [latex]\alpha = \Delta l[/latex].
Thus, the coefficient of linear expansion may be defined as the change in length to the per unit original length per unit change in temperature. Its value is different for different materials.
From eqn. (1), we get,
l2-l1 = [latex]\alpha l_1 \Delta \theta[/latex]
Or, l2 = l1 + l1[latex]\alpha \Delta \theta[/latex]
[latex]l_2 = l_1 (1+\alpha \Delta \theta)[/latex] …………………………… (3)
Unit of [latex]\alpha[/latex]
The unit of [latex]\alpha[/latex] is oC-1 or K-1.
Coefficient of Linear Expansion of a solid by Pullinger’s Apparatus
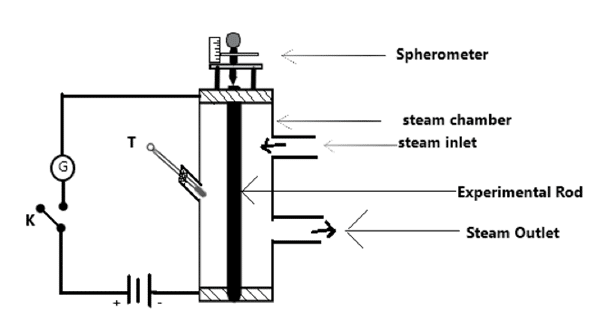
The experimental method to determine coefficient of linear expansion is as shown in figure. It consists of steam chamber having three holes for steam inlet, steam outlet and thermometer. An experimental rod is placed vertically at the middle of steam chamber. From the two ends of the rod, a galvanometer is connected along with the cell as shown in figure. And, spherometer is placed at the top of rod. The galvanometer shows deflection when central leg of spherometer touches the rod. Now, steam is passed from steam inlet and recorded the temperature by thermometer. And the expansion of metal rod is measured by spherometer.
Let,
Initial length of rod = l1
Initial temperature of rod = [latex]\theta_1^oC[/latex]
Final temperature of rod = [latex]\theta_2^oC[/latex]
Initial reading of spherometer = R1
Final reading of spherometer = R2
Now, from the definition of linear expansion
[latex]\alpha = \frac{Change\ in\ length}{Original\ length\ \times\ change\ in\ temperature}[/latex]
[latex]\alpha = \frac{R_2 – R_1}{l_1(\theta_2 – \theta_1)}[/latex]
This is the required expression for coefficient of linear expansivity from Pullinger’s apparatus.
Expansion of Liquid
Liquids, just like solids expands on heating & contracts on cooling. The expansion on liquid is more than that of the solid. It is because, the intermolecular force of attraction on liquid is less than that of solid. So, the molecules vibrate quickly on liquid than solid.
For the study of expansion of liquid, we have to study about the cubical expansion. We know, liquid have no shape but have definite volume. When it is kept in a vessel & then heated, the volume of liquid as well as the vessel increases.
Real & Apparent expansion of a liquid
The expansion of liquid on rise in temperature is always associated with the expansion of vessel. The expansion, which we observe is called apparent expansion. It is always less than real expansion of the liquid.
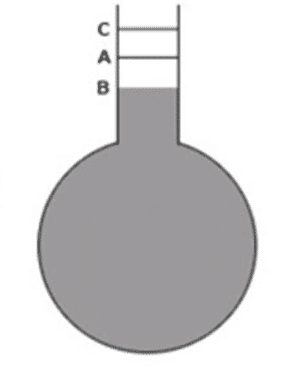
Let us take a round bottom flask made of glass with long narrow stem. Let, it be filled with liquid upto A. If the bulb is immersed in a hot liquid bath, the liquid level comes down to B. This is because the vessel is heated before the liquid. So, it expands first and the level of liquid decreases. After this, the liquid is heated and its level rises to C.
So, real expansion of liquid = increase in volume from B to C
Or, real expansion of liquid = increase in volume from B to A + increase in volume from A to C
Or, real expansion of liquid = expansion of vessel + apparent expansion of liquid
i.e., VBC = VAB + VAC ………………… (i)
Relation between Coefficient of Real and Apparent Expansion
Let us consider, a liquid contained in a vessel at room temperature [latex]\theta_1[/latex]. If V be the original volume, then, suppose, the liquid is heated to temperature [latex]\theta_2[/latex]. So,
Coefficient of real expansion of liquid [latex](\gamma_r) = \frac{Real\ increase\ in\ volume}{Original\ volume \times rise\ in\ temperature}[/latex]
= [latex]\frac{V_{BC}}{V\times \Delta \theta}[/latex]
[latex]V_{BC} = V\gamma_r \Delta \theta[/latex]
Also, coefficient of apparent expansion of liquid [latex](\gamma_a)[/latex]
= [latex]\frac{\text{Apparent increase in volume}}{Original\ volume \times rise\ in\ temperature}[/latex]
= [latex]\frac{V_{AC}}{V\times \Delta \theta}[/latex]
Or, VAC = [latex]V\gamma_a \Delta \theta[/latex]
Now, coefficient of volume expansion of glass vessel [latex](\gamma_g)[/latex]
= [latex]\frac{\text{Increase in volume of vessel}}{Original\ volume \times rise\ in\ temperature}[/latex]
= [latex]\frac{V_{AB}}{V\times \Delta \theta}[/latex]
Or, VAB = [latex]V\gamma_g \Delta \theta[/latex]
So, from equation (i),
VBC = VAB + VAC
Or, [latex]V\gamma_r \Delta = V\gamma_g \Delta \theta + V\gamma_a \Delta \theta[/latex]
Or, [latex]\gamma_r = \gamma_g + \gamma_a[/latex]
This is the required relation between coefficients of real and apparent expansivity of a liquid.
Determination of the co-efficient of real expansion of a liquid by Dulong and Petit’s method
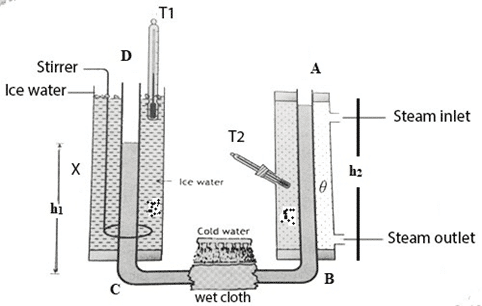
The apparatus consists of a U- shaped tube which contains liquid whose coefficient of real expansion is to be determined. The vertical limbs are surrounded by glass jackets. One jacket is placed with ice water and another jacket is placed at steam temperature by passing steam continuously through it. The horizontal part of the tube is continuously cooled by spraying cold water on it to prevent the flow of heat due to conduction across it. Ice water and steam is passed for a long time until it attains steady state separately and the temperatures are noted.
Calculation
Let, the temperatures of the cold and hot limbs be [latex]\theta_1^oC[/latex] and [latex]\theta_2^oC[/latex] respectively. For the liquid to be in steady state, the pressure at B must be equal to the pressure at C.
If h1 and h2 be the heights of the liquid columbs and [latex]\rho_1[/latex] and [latex]\rho_2[/latex] be the densities of the liquid at temperatures [latex]\theta_1^oC[/latex] and [latex]\theta_2^oC[/latex] respectively, then,
[latex]h_1\rho_1 g = h_2\rho_2 g[/latex] ………………….. (i)
[latex]\frac{h_1}{h_2} = \frac{\rho_2}{\rho_1}[/latex] ……………….. (ii)
But, density at [latex]\theta_2[/latex],
[latex]\rho_2 = \frac{\rho_1}{1+\gamma \Delta \theta}[/latex] ………………….. (iii)
And from eqn. (ii), we get,
[latex]\frac{h_1}{h_2} = \frac{\rho_1}{\rho_1(1+\gamma \Delta \theta)}[/latex]
Or, [latex]h_2 = h_1 + h_1 \gamma \Delta \theta[/latex]
[latex]\therefore h_2 – h_1 = h_1 \gamma \Delta \theta[/latex]
Or, [latex]\gamma = \frac{h_2 – h_1}{h_1\Delta \theta}[/latex]
By knowing the values of RHS, the coefficient of real expansion of liquid can be determined.