- A mixture of 500 g water and 100 g ice at 0oC is kept in a copper calorimeter of mass 200 g. How much steam from the boiler be passed to the mixture so that the temperature of the mixture reaches 40oC? (Latent heat of fusion of ice = 3.36 x 105 JKg-1, specific heat capacity of copper = 400 Jkg-1 oC-1, latent heat of vaporization of steam = 2.26 x 106 Jkg-1).
Solution
Given,
Mass of water (mw) = 500 g = 0.5 kg
Mass of ice (mi) = 100 g = 0.1 kg
Temperature [latex](\theta_1[/latex]) = 0oC
Mass of calorimeter (mc) = 200 g = 0.2 kg
Temperature of mixture [latex](\theta[/latex]) = 40oC
Mass of steam (ms) =?
We have, heat gained by ice, water and calorimeter
Qgained = (miLf + misw [latex]\Delta \theta[/latex]) + (mwsw [latex]\Delta \theta[/latex] + (mcsc [latex]\Delta \theta[/latex])
= (0.1 x 3.36 x 105 + 0.1 x 4200 x 40) + (0.5 x 4200 x 40) + (0.2 x 400 x 40)
= 1.376 x 105 J
Heat lost by steam
Qlost = (msLv + mssw )
= ms x 2.26 x 106 + ms x 4200 x (100 – 40)
= (2.26 x 106 + 2.52 x 105)ms
= 2.512 x 106ms
According to principle of calorimetry,
Qgained = Qlost
Or, 1.376 x 105 = 2.512 x 106ms
Or, ms = [latex]\frac{1.376\times 10^5}{2.512\times 10^6}[/latex]
= 0.05477 kg
= 54.77 gm.
- What is the result of mixing 100 g of ice at 0oC and 100 g of water at 100oC. Latent heat of fusion of ice = 336 x 103 JK-1, specific heat of water = 4200 Jkg-1K-1.
Solution
Given,
Mass of ice (mi) = 100 gm
Mass of water (mw) = 100 gm
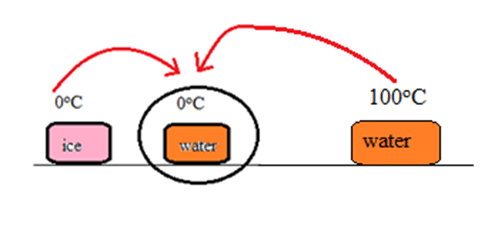
Amount of heat required to melt ice (Q1) = miLf
= 100 x 80
= 8000 Cal
Now, amount of heat lost by water and vessel i.e. amount of heat available for melting ice (Q2) = mwsw
= 100 x 1 x 100
= 10000 Cal
Since, Q2 > Q1, all ice melts, and the resulting temperature rises to [latex]\theta^oC[/latex]
Again, let’s have a look on the following figure.
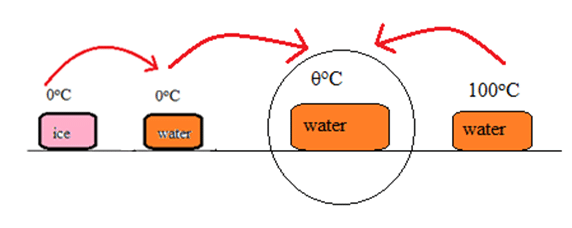
Heat gained by ice on changing from 0oC to [latex]\theta^oC[/latex],
Qgained = miLf + misw
= 100 x 80 + 100 x 1 x [latex]\theta[/latex]
= 8000 + 100[latex]\theta[/latex]
Heat lost by water from 100oC to [latex]\theta^oC[/latex],
Qlost = mwsw
= 100 x 1 x (100 -[latex]\theta[/latex])
= 10000 – 100[latex]\theta[/latex]
From the principle of calorimetry,
Heat lost = Heat gained
i.e. 10000 – 100 = 8000 + 100
Or, 10000 – 8000 = 100
Or, 2000 = 200[latex]\theta[/latex]
Or, [latex]\theta = \frac{2000}{200} = 10^oC[/latex]
So, in the mixture,
Mass of water formed = 100 + 100 = 200 gm
Mass of ice remaining = 100 – 100 = 0 gm
And, resulting temperature of mixture = 10oC.