MCQs
- [2082] What happens in an adiabatic process?
a. Volume remains constant
b. Pressure remains constant
c. Temperature remains constant
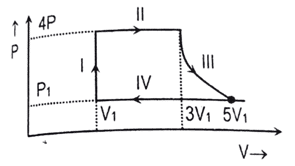
d. The system is insulated from the surroundings - [2081 GIE ‘A’] In the given PV diagram, which of the labelled parts of the cycle identify isobaric changes and adiabatic change of state?

| Options | Isobaric | Adiabatic |
| a | IV only | I & II only |
| b | I and II only | II and IV only |
| c | II and III only | IV only |
| d | I & III only | II only |
- [2081 GIE ‘B’] An inflated tyre of a bicycle burst. Which of the following relation between pressure P and temperature T holds good?
a. [latex]P^{1-\gamma}T^{\gamma – 1}[/latex]
b. [latex]P^{\gamma – 1}T^{\gamma – 1}[/latex]
c. [latex]P^{\gamma – 1}T^\gamma[/latex]
d. [latex]P^{1-\gamma}T^\gamma[/latex] - [2081 ‘B/C’] A system undergoes an isochoric process so that its temperature changes from 127oC to 227oC. What is the ratio of initial pressure to final pressure?
a. [latex]\frac{4}{5}[/latex]
b. [latex]\frac{5}{4}[/latex]
c. [latex]\frac{227}{127}[/latex]
d. [latex]\frac{127}{227}[/latex] - [2081 ‘D’] An ideal gas is taken through series of changes represented in diagram. The workdone by the gas at the end of cycle is
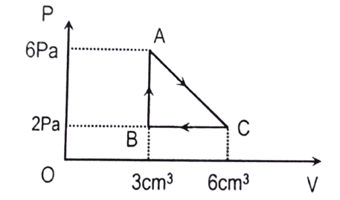
- [latex]6\times 10^{-6}\ J[/latex]
- [latex]12\times 10^{-6}\ J[/latex]
- [latex]3\times 10^{-6}\ J[/latex]
- 6 J
- [2080 GIE ‘A’] In which of the following processes of the gas, work done is maximum?
a. Isothermal
b. Adiabatic
c. Isobaric
d. Isochoric - [2080 GIE ‘B’] Internal energy of an ideal gas depends on
a. Temperature only
b. Pressure only
c. Volume only
d. Both pressure and volume - [2080 ‘P’] An ideal liquid is taken through series of changes represented in diagram. The work done by the gas at the end of cycle is
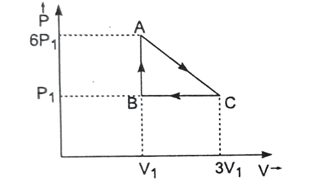
- [latex]P_1V_1[/latex]
- [latex]5P_1V_1[/latex]
- [latex]10P_1V_1[/latex]
- Zero
- [2080 ‘R’] An ideal gas is taken through series of changes represented in the diagram below. The net workdone by the gas at the end of the cycle is equal to
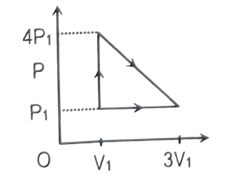
- [latex]P_1V_1[/latex]
- [latex]12P_1V_1[/latex]
- [latex]6P_1V_1[/latex]
- [latex]3P_1V_1[/latex]
- [2079 GIE ‘A’] A gas is initially at 27oC. It is compressed adiabatically from 27 litres to 8 litres. The rise in temperature is
a. 402oC
b. 375oC
c. 675oC
d. 537oC - [2079 GIE ‘B’] During adiabatic compression of a gas, its temperature
a. falls
b. remains constant
c. rises
d. becomes zero - [2079 ‘O’]Identify the wrong statement
a. for isothermal process, [latex]\Delta T = 0[/latex]
b. for isochoric process, [latex]\Delta V = 0[/latex]
c. for isobaric process, [latex]\Delta P = 0[/latex]
d. for cyclic process, [latex]\Delta w = 0[/latex] - [2079 ‘V’] An ideal gas ratio of heat capacities = 5/3 at 72oC is expanded adiabatically to eight times to its original volume. Approximate rise in temperature of the gas is
a. 86 K
b. 186 K
c. 259 K
d. 273 K
Answers
| 1.d | 2.d | 3.d | 4.d | 5.a | 6.c | 7.a | 8.b | 9.d | 10.b |
| 11.c | 12.d | 13.a |
THEORETICAL QUESTIONS
New Course
- [2082] State first law of thermodynamics. Does it follow the principle of conservation of energy? Explain. [2]
- [2081 GIE ‘A’] Why has a gas two specific heat capacities? Explain. [1+1]
- [2081 GIE ‘B’]
- Define isothermal process in thermodynamics. [1]
- Derive the relation [latex]C_p – C_v = R[/latex], where the symbols have their usual meanings. [3]
- Write the mathematical expression for entropy. [1]
- [2081 ‘B/C’] The given figure represents the P-V diagram of a cyclic process.
- Define indicator diagram. [1]
- Estimate the workdone during process. [2]
- Identify the thermodynamic process represented by I, II, III and IV. [2]
- [2081 ‘D’] Adiabatic process is the thermodynamic process in which the heat contained in a gaseous system remains constant.
- Adiabatic curve is much steeper than an isothermal curve, why? [1]
- Derive an expression for workdone by an ideal gas in an adiabatic process. [2]
- [2080 GIE ‘A’] Why is [latex]C-p[/latex] always greater than [latex]C_v[/latex] Explain. [2]
- [2080 GIE ‘B’]
- What is an isothermal process? [1]
- Derive an expression for work done by one mole of an ideal gas during isothermal expansion from volume [latex]V_1[/latex] to volume [latex]V_2[/latex]. [2]
- [2080 ‘P’] What is meant by entropy? Write the first law of thermodynamics in terms of entropy. [1+1]
- [2080 ‘R’]
- State first law of thermodynamics. [1]
- Apply the first law of thermodynamics to relate the two specific heat capacities of a gas. i.e. [latex]C_p – C_v = R[/latex]. [2]
- [2079 GIE ‘A’]
- What is an isothermal process? [1]
- Derive an expression for the workdone during isothermal process. [3]
- Can work be done by the system without changing its volume? [1]
- [2079 GIE ‘B’]
- State first law of thermodynamics. [1]
- [latex]C_p[/latex] is always greater than [latex]C_v[/latex], explain why? [2]
- [2079 ‘O’] Adiabatic process:
- Define adiabatic process in thermodynamics. [1]
- Derive expression for work done during adiabatic process. [3]
- Write the mathematical expression for entropy. [1]
- [2079 ‘V’] What happens to the energy added to an ideal gas when it is heated at?
- constant volume
- constant pressure [2]
Old Course
SHORT ANSWER QUESTION
- [2076 ‘GIE’ ‘A’] State and explain zeroth law.
- [2076 ‘GIE’ ‘A’] A certain quantity of ideal gas is compressed to half of its initial volume. The process may be adiabatic or isothermal. For which process greater amount of mechanical work is required?
- [2076 ‘GIE’ ‘B’] What are the limitations of first law of thermodynamics?
- [2076 ‘B’] The internal energy of a compressed gas is less than that of a rarefied gas at the same temperature. Why?
- [2076 ‘C’] Why does the temperature of a gas drop during an adiabatic expansion?
- [2075 ‘GIE’] Why does the temperature of a gas undergoing adiabatic expansion decrease?
- [2075 ‘A’] Why does a gas have two specific heat capacities?
- [2074 ‘B’] Explain the significance of the first law of thermodynamics. Hence write its expression in terms of the change in entropy of the system.
- [2073 ‘D’] Compare the internal energy of a compressed gas with that of the rarefied gas at the same temperature.
- [2072 ‘C’] Why specific heat capacity of gas at constant pressure is greater than its specific heat capacity at constant volume?
- [2072 ‘D’] When a gas expands adiabatically, it does work on its surroundings. But if there is no heat input to the gas where does the energy come from?
LONG ANSWER QUESTION
- [2076 ‘GIE’ ‘A’] Why a gas has two values of specific heat capacity? Show that for an ideal gas Cp – CV = R where symbols have their usual meanings.
- [2076 ‘GIE’ ‘B’] For an adiabatic process, show that [latex]PV^\gamma[/latex] = constant, where symbol have their usual meanings.
- [2076 ‘C’] State and explain first law of thermodynamics. Derive the relation, CP – CV = R, where the symbols have their usual meanings.
- [2075 ‘A’] What do you mean by adiabatic process? Deduce the equation [latex]PV^\gamma[/latex] = constant, where the symbols have their usual meaning.
- [2074 ‘S’] Why does a gas have two molar heat capacities? Show that for an ideal gas; Cp – Cv = R, where symbols have their usual meanings.
- [2074 ‘A’] Why does a gas have two molar heat capacities? Derive relation between them for an ideal gas.
- [2074 ‘B’, 2073 ‘C’, 2073 ‘E’] What is the difference between isothermal and adiabatic process? Prove that [latex]PV^\gamma[/latex] is constant in an adiabatic process, [latex]\gamma[/latex] is the ratio of Cp and Cv.
- [2073 ‘S’] What is adiabatic process? Derive an expression for the workdone in this process.
- [2073 ‘C’] What is an isothermal process? Derive the relation, [latex]PV^\gamma[/latex] = constant, where symbols have their usual meanings.
- [2072 ‘D’] Explain the term, thermodynamic process. Obtain an expression for the work done by a gas during adiabatic expansion.
NUMERICAL PROBLEMS
New Course
- [2080 ‘P’] A quantity of monoatomic gas at 30oC is compressed suddenly to ([latex]\frac{8}{27}[/latex]) parts of its initial volume. Find the change in temperature assuming [latex]\gamma (=\frac{C_p}{C_v})[/latex] to be [latex]\frac{5}{3}[/latex]. [2] Ans: 379 K
Old Course
- [2076 ‘B’] A certain volume of dry air at NTP is allowed to expand five times of its original volume under adiabatic condition. Calculate the final pressure and temperature. ([latex]\gamma = 1.4[/latex]). Ans: 1.06 x 104 Nm-2 (0.105 atm), 143.4 K
- [2075 ‘S’] A certain volume of dry air at NTP is allowed to expand four times of its original volume under (i) isothermal conditions (ii) adiabatic conditions. Calculate the final pressure and temperature in each case. ([latex]\gamma = 1.4[/latex]). Ans: (i) 2.5 x 104 Nm-2 (0.25 atm), 273 K (ii) 1.45 x 104 Nm-2 (0.143 atm), 156.8 K
- [2075 ‘B’] A gas in a cylinder initially at a temperature of 10oC and one atmospheric pressure is to be compressed adiabatically to 1/8 of its volume. Find the final temperature. (Take [latex]\gamma[/latex] = 1.4). Ans: 650.2 K
- [2073 ‘D’] A gas in a cylinder is initially at a temperature of 17oC and pressure 1.01 x 105 Nm-2. If it is compressed adiabatically to one-eighth of its original volume, what would be the final temperature and pressure of the gas? Ans: 666.2 K, 1.85 x 106 Nm-2
- [2072 ‘S’] For hydrogen, the molar heat capacities at constant volume and constant pressure are 20.5 Jmol-1K-1 and 28.8 Jmol-1K-1. Calculate
- The heat needed to raise the temperature of 8g of hydrogen from 10oC to 15oC at constant pressure.
- The increase in internal energy of the gas. Ans: 576 J, 410 J
- [2072 ‘C’] Five moles of an ideal gas are kept at constant temperature of 53oC while the pressure of the gas is increased from 1.00 atm to 3.00 atm. Calculate work done on the gas. Ans: – 1.49 x 104 J
- Air is compressed adiabatically to half its volume. Calculate the change in its temperature. Ans: 32%
- A monoatomic ideal gas that is initially at a pressure of [latex]1.50\times 10^5[/latex] pa and has a volume of 0.08 m3 compressed adiabatically to a volume of 0.04 m3. (a) What is the final pressure? (b) How much work is done by the gas? (c) What is the ratio of the final temperature of the gas to its initial temperature? Ans: 476220 pa; 10573 J, 1.58
- If the ratio of specific heat capacities of a gas is 1.4 and its density at S.T.P. is 0.09 kg/m3. Calculate the value of specific heat capacities at constant pressure and at constant volume. Ans: [latex]1.03\times 10^4\ Jkg^{-1}K^{-1}[/latex], [latex]1.44\times 10^4\ Jkg^{-1}K^{-1}[/latex]
- Gas in a cylinder initially at a temperature of 17oC and pressure [latex]1.01\times 10^5\ Nm^{-1}[/latex] of is to be compressed adiabatically to one-eight of its volume. Find the temperature and pressure of the gas. Ans: [latex]1.85\times 10^6\ N/m^2[/latex] 666. 24 K
- A litre of air, initially at 20oC and at 760 mm of Hg pressure, is heated at constant pressure until its volume is doubled. Find (i) the temperature, (ii) external work done by the air in expanding, and (iii) the quantity of heat supplied. Specific heat capacity at constant volume = 714 J/kg/K. Ans: 586 K, 101.3 J, 352.8 J
- An ideal gas initially at 4 atmosphere and 300 K is permitted to expand adiabatically twice its initial volume. Find the final pressure and temperature if the gas is (i) monoatomic and (ii) diatomic with [latex]C_v[/latex] = 5/2R. Ans: (a) 1.257 atm; 189K (b) 1.52 atm; 227.35 K
- The density of an ideal gas is 1.6 [latex]kgm^{-3}[/latex] at 27oC and 105 [latex]Nm^{-2}[/latex] pressure. Its specific heat capacity at constant volume is 312 [latex]JKg^{-1}K^{-1}[/latex]. Find the ratio of the specific heat at constant pressure to that at constant volume. Ans: 1.67
- The density of a gas is 1.775 [latex]Kgm^{-3}[/latex] at 27oC and 105 [latex]Nm^{-2}[/latex] pressure. If the specific heat capacity at constant pressure is 846 [latex]JKg^{-1}K^{-1}[/latex]. Find the ratio of specific heat capacity at constant pressure to that at constant volume. Ans: 1.29
- An ideal gas is slowly compressed at constant temperature of 50oC to one half of its original volume. In this process, 80 cal of heat was given. How much work was done and what was the change in the internal energy of the gas? Assume one mole of an ideal gas. Ans: 807.03 J; 1143.03 J
- 16 g of oxygen having volume 0.02 m3 at a temperature of 27oC and pressure [latex]2\times 10^5\ Nm^{-2}[/latex] is heated at constant pressure until its volume increases to 0.03 m3. Calculate the external work done and increase in internal energy of the gas if its molar heat capacity at constant volume is 0.8 [latex]Jmol^{-1}K^{-1}[/latex] and molar mass of oxygen is 32. Ans: [latex]2\times 10^3[/latex] J, 60 J
- A gasoline engine takes in air at 25oC and one atmospheric pressure and compresses adiabatically to one-tenth of its original volume. Find the final temperature and pressure. [latex](\gamma = 1.4)[/latex]. Ans: 748.5 K, 25.11 atmosphere
- A mass of air occupying initially a volume [latex]2\times 10^{-3}\ m^{-3}[/latex] at a pressure of 760 mm of mercury and a temperature of 20oC is expanded adiabatically and reversibly to twice its volume, and then compressed isothermally and reversibly to a volume of [latex]3\times 10^{-3}[/latex] m3. Find the final pressure assuming the ratio of the specific heat capacities of air to be 1.4. Ans: 384 mm of Hg
- Gas in a cylinder, initially at a temperature of 10oC and pressure of [latex]1.01\times 10^5\ Nm^{-2}[/latex] is to be compressed adiabatically to one eight of its volume. Find final pressure and temperature. (Ratio of molar heat capacities = 1.40) Ans: [latex]1.85\times 10^5\ Nm^{-2}[/latex], 560.16 K