MCQs
- [2079 ‘O’] Which of the following one is correct?
a. E2 = P2C
b. E2 = P2C2
c. E2 = PC2
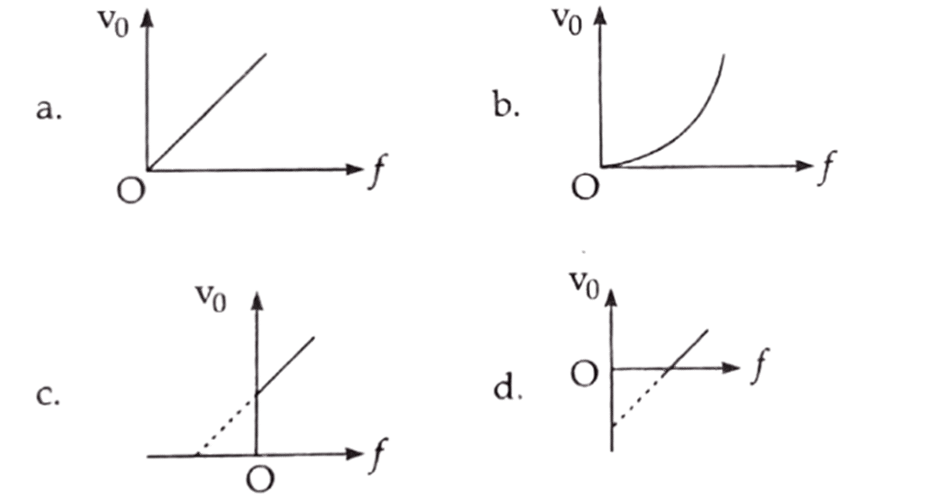
d. E2 = P2/C2 2. - [2079 ‘V’] In photoelectric cell, the relation between cut-off voltage (Vo) and frequency (f) of incident light is best represented by
THEORETICAL QUESTIONS
New Course
- [2082] Define threshold wavelength. [1]
- [2081 GIE ‘B’]
- Is photoelectric equation possible for all wavelengths of light? Justify. [2]
- Write Einstein’s photo electric equation and point out any two characteristic properties of photons. [2]
- Photons come from three lamps that emit red, green and blue light. Which of these lamps produces photons with the highest energy? [1]
- [2081 ‘B/C’] Explain photoelectric effect and write Einstein’s photoelectric equation. [2]
- [2081 ‘D’] Sketch the graphs, showing the variation of stopping potential (Vo) with the frequency of incident radiation for two photosensitive materials A and B having threshold frequency fo and fo’ such that fo > fo’. [1]
- Which of two materials A or B has higher work junction? [1]
- What does the value of intercept of graph A on the potential axis gives? [1]
- [2080 GIE ‘B’] When introducing optoelectronics to a class, a physics teacher writes “One of the important factors affecting photoelectric emission from a metal is the threshold frequency for the metal.”
Define the terms:
- Photoelectric emission. [1]
- Threshold frequency. [1]
- [2080 ‘P’] A graph between stopping potential (Vo) with the frequency (f) of incident radiation is shown in figure below. Answer the following questions.
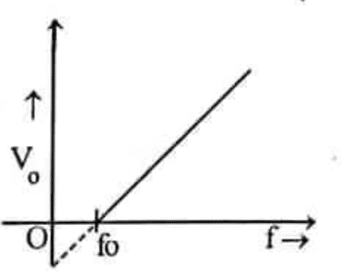
- Write the Einstein photoelectric equation in terms of stopping potential. [1]
- How will you determine Planck’s constant from the graph? [2]
- What happens to stopping potential if frequency of incident photon increases gradually? Justify. [2]
Old Course
SHORT ANSWER QUESTION
- [2076 ‘B’] Which has more energy – a photon in the infrared or photon in the ultraviolet? Give reasons.
- [2076 GIE ‘A’] Can photoelectric effect be explained on the basis of wave theory of light? Justify your answer. [2]
- [2075 GIE] If the wavelength of electromagnetic radiation is doubled, what will happen to the energy of the photons?
- [2075 ‘A’]What happens to the kinetic energy of photoelectrons when the intensity of incident light is doubled? Justify your answer.
- [2072 ‘S’]Human skin is relatively insensitive to visible light, but ultra violet radiation can cause severe burns. Does this have anything to do with photon energies? Explain.
- [2072 ‘C’]If we go on increasing the wavelength of light incident on a metal surface, what charges take place in the number of electrons of energy of electrons?
LONG ANSWER QUESTION
- [2078 ‘C’] Define photoelectric effect and describe Einstein’s photoelectric equation. Explain the terms-threshold frequency and work function.
- [2076 ‘C’]What is photoelectric effect? Discuss Einstein’s photoelectric equation. Write some applications of photoelectric effect.
- [2074 ‘S’]What is photoelectric effect? Explain the effect of increase of (i) frequency (ii) intensity of the incident radiation on photoelectrons emitted by a phototube.
- [2074 ‘A’, 2072 ‘D’]Discuss photoelectric effect and derive Einstein’s photoelectric equation. What is stopping potential?
- [2075 ‘S’, 2073 ‘C’]Explain photoelectric effect to write Einstein’s photoelectric equation. Describe Millikan’s laboratory method to determine Planck’s constant.
OR [2072 ‘E’] Explain Millikan’s experiment for the verification of Einstein’s photoelectric equation.
NUMERICAL PROBLEMS
New Course
- [2082] A metal has a work function of 2 eV. Calculate the maximum speed of the emitted photoelectrons when it is illuminated by radiation of wavelength 150 nm. What is the least frequency of the radiation for which electrons are emitted? (Given: Planck’s constant h = [latex]6.62\times 10^{-34}\ Js,\ m = 9.1\times 10^{-31}\ Kg, e = 1.6\times 10^{-19}\ C[/latex]) [3] Ans: [latex]1.48\times 10^6[/latex] m/s
- [2080 GIE ‘A’] The photon is then incident on the silver surface in a photoelectric experiment and the surface emits an electron with maximum possible kinetic energy. If the work function of silver is 4.7 eV,
- What do you mean by work function? [1]
- Calculate the maximum kinetic energy of the emitted electron. [1]
- Determine the stopping potential for the emitted electron. [1] Ans: (ii) 5.56 eV (iii) 5.56 V
- [2080 GIE ‘B’] The minimum energy required to cause an electron to be emitted from a clean Zinc surface is [latex]6.9\times 10^{-19}\ J[/latex].
- Calculate the maximum wavelength of electromagnetic radiation which will cause an electron from the Zinc surface. [2]
- What would be the effect of irradiating the Zinc surface with radiation of wavelength [latex]4\times 10^{-7}[/latex] m? Justify your answer. [2] Ans: (i) [latex]2.88\times 10^{-7}[/latex] m (ii) No photo electrons are emitted
- [2080 GIE ‘B’] When light of wavelength [latex]\lambda_1[/latex] is incident on the cathode of a photoelectric tube, the maximum kinetic energy of the emitted electron is 1.8 eV. If the wavelength is reduced to [latex]\frac{\lambda_1}{2}[/latex], the maximum kinetic energy of the emitted electron is 5.5 eV. Find the work function of the cathode materials. [2] Ans: 1.9 eV
- [2080 ‘R’] In an experiment on photoelectric effect, the graph between maximum kinetic energy and frequency of incident radiation on the metal surface is found to be straight line as shown in the figure.
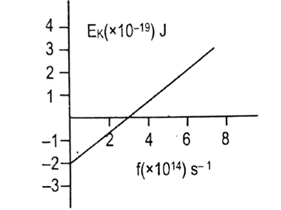
Calculate:
- threshold frequency. [1]
- Planck’s constant. [1]
- work function of metal in eV. [1] Ans: (i) [latex]3\times 10^{14}\ s^{-1}[/latex] (ii) [latex]6.67\times 10^{-34}\ Js[/latex] (iii) 1.25 eV
- [2079 GIE ‘B’]
- What are photons? [1]
- The adjacent figure shows a plot of stopping voltage versus frequency of incident radiation for photoelectric effect in sodium.
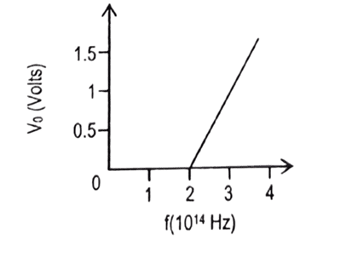
- Calculate the threshold frequency and work function of sodium. [2]
- If intensity of the incident radiation is increased, does the slope of the line change? Justify your answer. [2] Ans: (i) [latex]2\times 10^{14}\ Hz[/latex], 0.83 eV
- [2079 ‘V’]
The work function of a metal is [latex]6.4\times 10^{-19}\ J[/latex].
- Explain what is meant by the term ‘work function’. [2]
- Light with a frequency of is shone onto the metal surface. Find whether or not the photons of this light will cause the photoelectric effect to take place. [latex](h = 6.62\times 10^{-34}\ Js)[/latex]
- The light source is now replaced with a light source of frequency [latex]1.5\times 10^{15}\ Hz[/latex].
- Write down Einstein’s photoelectric equation. [1]
- The photons from the source contain more energy than is required to release the electrons. How much extra energy is available after the electron has been released? [2]
- Photons come from three lamps that emit red, green and blue light. Which of these lamps produce photons with the highest energy? [1] Ans: [latex]5.53\times 10^{-19}\ J[/latex]
Old Course
- [2076 GIE ‘B’] Find the change in stopping potential when the wavelength of a radiation is decreased from 400 nm to 300 nm on the same metallic surface. Planck’s constant = [latex]6.62\times 10^{-34}\ Js[/latex] and velocity of light = [latex]3\times 10^8\ m/s[/latex]. Ans: 1.034 V
- [2075 ‘A’] A clean nickel surface of work function 5.1 eV is exposed to light of wavelength 235 nm. What is the maximum speed of the photoelectrons emitted from its surface? Ans: 2.52 x 105 m/s
- [2075 ‘B’] Sodium has a work function of 2eV. Calculate the maximum energy and speed of the emitted electrons when sodium is illuminated by a radiation of 150 nm. What is the threshold frequency of radiation for which electrons are emitted from sodium surface? Ans: 10-18 J, 1.5 x 106 m/s, 4.8 x 1014 Hz
- [2074 ‘B’] Radiations of wavelength 5400 Å fall on a metal whose work function is 1.9 eV. Find the energy of the photoelectrons emitted and their stopping potential. (Planck’s Constant = 6.62 x 10-34 Js). Ans: 0.39 eV, 0.39 V
- [2073 ‘S’] Light of frequency 5 x 1014 Hz liberates electrons with energy 2.3 x 10-19 J from a certain metallic surface. What is the wavelength of ultraviolet light which liberates electrons of energy 8.93 x 10-19 J from the same surface? (Given h = 6.62 x 10-34 Js, c = 3 x 108 m/s). Ans: 2 x 10-7 m
- [2073 ‘D’] 400 nm wavelength of light falls on a photo sensitive material of work function 2.3 eV. Compute the maximum energy of photoelectrons. Ans: 0.803 eV
- [2072 ‘S’]Sodium has a work function of 2eV. Calculate the maximum energy and speed of the emitted electrons when sodium is illuminated by radiation of wavelength 150 nm. (Given mass of electron = 9.1 x 10-31 kg). Ans: 1 x 10-18 J, 1.48 x 106 m/s
- [2072 ‘C’] The work function for the surface of aluminium is 4.2 eV. How much potential difference will be required to stop the emission of maximum energy electrons emitted by light of wavelength 2000 Å? (Planck’s constant = 6.6 x 10-34 Js). Ans: 1.98 V