MULTIPLE CHOICE QUESTIONS
- [2082] The efficiency of Carnot engine operating between steam point and ice point is
a. 26.8 %
b. 50%
c. 73.2%
d. 100%
- [2081 GIE ‘A’] An ideal refrigerator is working between 0oC and 127oC. The coefficient of performance is
a. 3.25
b. 2.85
c. 2.15
d. 1
- [2081 GIE ‘B’] What is the efficiency of a carnot engine working between steam point and ice point?
a. 27%
b. 37%
c. 50%
d. 75%
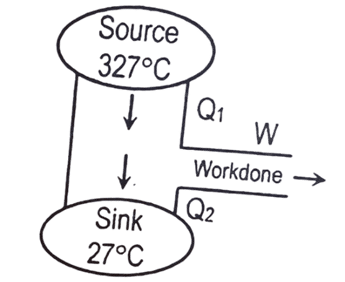
- [2081 B/C] Given figure represents the block diagram of a heat engine. What is the efficiency of this engine?
a. 25%
b. 50%
c. 60%
d. 75%
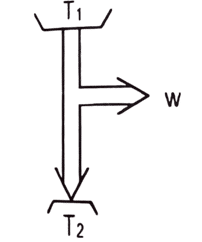
- [2081 D] An ideal heat engine working between temperatures T1 and T2 has efficiency [latex]\eta[/latex]. If both the temperature are raised by 50 K each, the new efficiency of engine will be
a. [latex]\eta[/latex]
b. more than [latex]\eta[/latex]
c. Less than [latex]\eta[/latex]
d. depends upon the nature of working substance
- [2080 GIE A] When the door of a refrigerator is left open in a room, the temperature of the room
a. Increases
b. Decreases
c. Remains same
d. First increases, then decreases
- [2080 GIE B] An ideal refrigerator is working between 0oC and 127oC. The coefficient of performance is
a. 1
b. 2.15
c. 2.85
d. 3.15
- [2080 P] A Carnot engine is working between 300 K and 600 K has output 200 J. What is the energy supplied to the engine?
a. 400 J
b. 800 J
c. 1200 J
d. 1600 J - [2080 R] What is the significance of the Clausius statement of the second law of thermodynamics?
a. Defines the maximum theoretical efficiency of a heat engine.
b. States that heat cannot be spontaneously flow from a colder object to a hotter object.
c. Describes the relationship between entropy and the reversibility of a process.
d. Describes the direction of spontaneous change in an isolated system. - [2079 GIE A] The efficiency of a carnot engine working between steam point and ice point is about.
a. 56.8% b. 44.8% c. 26.8% d. 14.8% - [2079 GIE B] The efficiency of a carnot engine working between 27oC and 127oC is
a. 50% b. 100% c. 25% d. 75% - [2079 ‘O’] The maximum efficiency of an engine operating between 30oC and 300oC is,
a. 4.71% b. 47% c. 90% d. 9% - [2079 ‘V’] A carnot cycle includes
a. Two isothermal and two adiabatic processes.
b. Two isothermal and two isobaric processes.
c. Two isothermal processes.
d. Two adiabatic processes.
Answers:
| 1.a | 2.c | 3.a | 4.b | 5.c | 6.a | 7.b | 8.a | 9.b | 10.c |
| 11.c | 12.b | 13.a |
THEORETICAL QUESTIONS
- [2080 GIE A] Draw a PV diagram of a petrol engine and explain its working based on the PV diagram. [3]
- [2080 P] Can a room be cooled by leaving the door of an electric refrigerator open? Justify. [1]
- [2080 R] Obtain the efficiency of a heat engine. [2]
Old Course
SHORT ANSWER QUESTION
- [2074 ‘S’] Why does a refrigerator consume more power in summer than in winter to cool the same quantity of food by same degree?
- [2074 ‘A’] Why not an ideal heat engine is cent percent efficient?
- [2073 ‘S’] An engine cannot have 100% efficiency. Why and which basic expression of thermodynamic law suggest this?
- [2073 ‘C’] Explain the significance of second law of thermodynamics.
- [2072 ‘A’] Entropy is defined in the second law of thermodynamics. Can the first law be expressed in terms entropy? How?
LONG ANSWER QUESTION
- [2076 ‘B’] Describe the working principle of diesel engine with the help of PV diagram.
- [2075 ‘GIE’] State and explain second law of thermodynamics. Define the efficiency of a heat engine and explain quantitatively why the efficiency of such an engine is always less than 100%?
- [2075 ‘B’] Describe the working principle of petrol engine with the help of its PV diagram.
- [2073 ‘D’] What is efficiency of a heat engine? Describe the working of a petrol engine with the help of its P-V diagram.
- [2072 ‘S’] Describe the working of a petrol engine with the help of its P-V diagram.
- [2072 ‘C’] Describe the working of diesel engine with a P-V diagram. What are its merits and demerits?
NUMERICAL PROBLEMS
New Course
- [2082] A Carnot’s engine has 25% efficiency with a sink at 9oC. By how many degrees should the temperature of the source be increased in order to raise the efficiency to 50%? [3] Ans: 188 K
- [2081 GIE ‘A’] For a carnot ideal engine, temperature of sink at temperature 27oC and source is at temperature 127oC. Calculate its efficiency. [3] Ans: 25%
- [2081 D] A refrigerator has a coefficient of performance of 1.95. In each cycle, it absorbs [latex]3\times 10^4[/latex] J of heat from cold reservoir. How much heat is discarded to the high temperature during each cycle? [2] Ans: [latex]4.5\times 10^4\ J[/latex]
- [2080 GIE B] Two carnot engines A & B have their sources at 400 K and 350 K, and sinks at 350 K and 300 K respectively. Which engine is more efficient and by how much? [2] Ans: B is more efficient than A by 1.78%
- [2079 GIE B] A carnot engine takes [latex]4.2\times 10^6\ J[/latex] of heat from reservoir at 627oC and performs external work. The remaining energy is rejected into a sink at 27oC. What is the efficiency? How much work does it perform? [1 + 1] Ans: [latex]2.8\times 10^6\ J[/latex]
- [2079 ‘V’] A Carnot engine working between 300 K and 600 K has a working output of 800 J per cycle. What is the amount of heat energy supplied to the engine by the source per cycle? [3] Ans: 1600 J/cycle
Old Course
- [2076 GIE A] A carnot engine takes 103 calories of heat from a reservoir at 227oC and rejects heat to a reservoir at 27oC. How much work is done by it? Ans: 1680 J
- [2076 GIE B] A diesel engine performs 2500 J of mechanical work and discards 4000 J of heat each cycle.
i. How much heat must be supplied to the engine each cycle?
ii. What is the thermal efficiency of the engine? Ans: 6500 J, 38.46% - [2076 ‘C’] The efficiency of a Carnot cycle is 15%. If on reducing the temperature of sink by 65oC, the efficiency becomes double, find the temperature of source and sink. Ans: 433.3 K, 368.3 K
- [2075 ‘A’] A carnot engine has 40% efficiency with sink at 10oC. By how many degrees should the temperature of the source be increased in order to raise the efficiency to 65%? Ans: 336.9 K
- [2074 ‘S’] A Carnot engine has 50% efficiency with a sink at 9oC. By how many degrees should the temperature of source be increased in order to raise the efficiency to 70%? Ans: 376K
- [2074 ‘A’] An ideal heat engine operates between two reservoirs at two temperatures. In order to achieve 30% efficiency when the temperature of the sink is 50oC, what should be the temperature of the source? Ans: 461.43 K
- [2074 ‘B’] The source reservoir of a carnot engine is at a temperature of 400 K and takes 400 J of heat and rejects 20 J of heat to the sink reservoir in each cycle. What is the efficiency of engine and the temperature of the sink? Ans: [latex]\eta = 95\%,\ T_2\ =\ 20K[/latex]
- [2073 ‘C’] A Carnot engine has 25% efficiency with a sink at 9oC. By how many degrees should the temperature of the source be increased in order to raise the efficiency to 70%? Ans: 564 K
- [2072 ‘D’] A diesel engine performs 2200 J of mechanical work and discards 4300 J of heat each cycle. (i) How much heat must be supplied to the engine in each cycle? (ii) What is the thermal efficiency of the engine? Ans: 6500 J, 33.85%
- [2072 ‘E’] What will be the thermal efficiency of an engine if it takes 8 KJ heat from the source and rejects 6 KJ to the sink in one cycle? Ans: 25%
Finished studying this chapter resource?
Mark this chapter as complete to update your course progress.